Professional Documents
Culture Documents
P-Ni Case Study
Uploaded by
bars20060 ratings0% found this document useful (0 votes)
26 views6 pagesP-Ni electroless deposits are used as a finish on many substrates. A clear, thin, dark line was observed to be developing at the interface between the Ni and Ni-Sn IMC layers. This interface is usually not very easily distinguishable initially after soldering to Ni deposits.
Original Description:
Copyright
© Attribution Non-Commercial (BY-NC)
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentP-Ni electroless deposits are used as a finish on many substrates. A clear, thin, dark line was observed to be developing at the interface between the Ni and Ni-Sn IMC layers. This interface is usually not very easily distinguishable initially after soldering to Ni deposits.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
26 views6 pagesP-Ni Case Study
Uploaded by
bars2006P-Ni electroless deposits are used as a finish on many substrates. A clear, thin, dark line was observed to be developing at the interface between the Ni and Ni-Sn IMC layers. This interface is usually not very easily distinguishable initially after soldering to Ni deposits.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 6
Case Study
Ref: P-Ni Failures
Attached is a partial copy of some work we conducted at Alpha as a result of a large
number of failure analyses we determined were related to electroless Ni deposits. Over the
past few years we have seen many failures that appeared to indicate a relationship of the
failures to P-Ni electroless deposits used as a finish on many substrates. It was my initial
hypothesis that the P was accumulating at the interface between the electroless P-Ni and the
Ni-Sn intermetallic compound (IMC) layer being formed with the Sn in the solder. My reasoning
being that since the P in the Ni deposit was not taking part in the reaction with the solder, it
would be accumulating somewhere in the microstructure of the solder connection. After
looking at hundreds of metallographically prepared cross-sections of failures where P-Ni was
involved, I began to see some clues that supported the proposed hypothesis. All of the failures
observed were taking place at the interface between the Ni and Ni-Sn IMC layers. A clear, thin,
dark line was observed to be developing at the interface between the Ni and Ni-Sn IMC. This
interface is usually not very easily distinguishable initially after soldering to Ni deposits,
especially the electrolytic Ni deposits which do not possess occluded P. As our knowledge base
grew, we saw more of the same evidence in our investigations of other failures of this type. In
several instances we were fortunate to have very severe widespread failures on many
assemblies submitted by particular sources.
In these instances, we were able to observe failures at almost every stage of
development. We saw interfaces with no distinct interfacial darkening that I was relating to the
P build-up. We also observed various stages of darkening where the interface between the Ni
and Ni-Sn IMC was becoming distinctly clear. And finally, we observed the various stages of
separation along with complete failure of the connections.
Subsequently, we examined several samples using an SEM with X-ray analysi s
capabilities and were able to confirm our hypothesis. We were also fortunate to have some
complete assemblies with a good number of BGA components that were failing electrical
testing. The motherboard to which the BGA's were attached was coated with electroless P-Ni
and immersion Au.
Metallographically prepared cross-sections of various BGA's on this assembly revealed
the various conditions that I've described in the previous text herein. All stages of the failure
development were observed, from no indication of interfacial darkening that we suspected to
be P, to complete separation and total failure of the connections. To confirm our P buildup
hypothesis, we took a portion of the assembly that had not been used for metallographic
analyses and carefully and deliberately lifted a couple of BGA components from the
motherboard by mechanical means to examine the failure surfaces. In particular, we were
interested in the failures at the Ni interface on the motherboard where the P-Ni coating was
present. We then took the two (2) mating BGA parts (BGA components and their Motherboard
attachment sites) and conducted analyses of a number of failed surfaces on both sides. The P
readings on the board side failure surfaces were discovered to be much higher than those on
the detached solder ball failure surfaces. Therefore, we began to survey a large number of
board side failure surfaces and compared the P values to the values determined by our
analyses of the P-Ni layers taken on the metallographic cross-sections.
We discovered that the average value of all the P measurements taken on the board
side failed surfaces were ~44%P. The average value of the measurements taken on the P-Ni
cross-sections was ~13%. The selected acceleration potential of the incident beam used to
make these determinations was 10keV. We determined this, by experimentation, to produce
reasonable surface readings with a minimal effect of the underlying P-Ni coating. The
anlayses, on the whole, indicate P is definitely present at the failure surfaces, primarily
adherent to the P-Ni surface, with lower levels on the Ni-Sn IMC surface on the detached solder
balls.
It is a fact that P-Ni coatings are usually more solderable than electrolytically applied
coatings, as claimed by the suppliers of plating solutions and react more quickly with the
solders to form IMC layers at their interfaces. Unfortunately, this beneficial characteristic
becomes one that is detrimental to reliability when the assemblies are subjected to multiple
soldering operations and other thermal excursions in service. When this occurs, the continued
diffusion between the Sn in the solder and the P-Ni coating results in a weakened bond
between the P-Ni and the Ni-Sn IMC reaction layer as the P concentration becomes elevated at
this interface.
We usually recommend that the P level of the deposit be kept as low as possible
without too badly impairing the beneficial properties imparted by its presence in the Ni deposit.
If the P level is too low, there is a risk of corrosion when applying the immersion Au coating
over the P-Ni. If it is too high, the risk of premature P buildup and failures is increased. There
are always trade-offs. Control of the P levels is usually accomplished by adjusting and
controlling some of the critical plating solution parameters.
I have attached some photomicrographs that illustrate many of the observations we've
made over the course of the last few years relative to this topic. I hope this background and the
associated photomicrographs are some help to you.
If any questions and/or comments arise as you consider these findings, please feel free
to contact me so that we may address them together.
Sincerely,
Ron Bulwith
rbulwith@alphametals.cookson.com (E-mail)
800-367-5460 (Toll Free)
201-324-3650 (Direct Dial)
201-324-3676 (Fax)
www.alphametals.com (Website)
You might also like
- Copper Electrodeposition for Nanofabrication of Electronics DevicesFrom EverandCopper Electrodeposition for Nanofabrication of Electronics DevicesNo ratings yet
- ASB Magazin, Oct 2011Document5 pagesASB Magazin, Oct 2011emediageNo ratings yet
- Materials Science and Engineering BDocument7 pagesMaterials Science and Engineering Beid elsayedNo ratings yet
- A Novel Method To Produce PD Nanoparticle Ink For Ink-Jet Printing TechnologyDocument5 pagesA Novel Method To Produce PD Nanoparticle Ink For Ink-Jet Printing Technologytpjoshi1No ratings yet
- Electro-Optical and Cathodoluminescence Properties of Low Temperature Grown Zno Nanorods/P-Gan White Light Emitting DiodesDocument6 pagesElectro-Optical and Cathodoluminescence Properties of Low Temperature Grown Zno Nanorods/P-Gan White Light Emitting DiodesHuynh Ngoc TienNo ratings yet
- Solar Cells IBCDocument6 pagesSolar Cells IBCYaroslav VainshteinNo ratings yet
- Wafer BondingDocument31 pagesWafer BondingNilesh SarkarNo ratings yet
- The Root Cause of Black PadDocument5 pagesThe Root Cause of Black PadxerxeshakaNo ratings yet
- Guard RingsDocument5 pagesGuard RingsnarendraNo ratings yet
- SAC BGAs in SNPB PDFDocument17 pagesSAC BGAs in SNPB PDFZetocha MilanNo ratings yet
- Impact of Printed Wiring Board CoatingsDocument10 pagesImpact of Printed Wiring Board CoatingsTin NguyenNo ratings yet
- Sic Mos Trench Self Align Micromachines-14-02212Document12 pagesSic Mos Trench Self Align Micromachines-14-02212terry chenNo ratings yet
- Evaluation of Pb-Free BGA Solder Joint ReliabilityDocument12 pagesEvaluation of Pb-Free BGA Solder Joint ReliabilityZetocha MilanNo ratings yet
- Pun2017 Article Solid-stateGrowthKineticsOfInt (3390)Document13 pagesPun2017 Article Solid-stateGrowthKineticsOfInt (3390)tl xNo ratings yet
- Guard RingsDocument10 pagesGuard RingsZekeZer0xNo ratings yet
- Led PowerpointDocument35 pagesLed PowerpointArunkumarNo ratings yet
- Junctionless CMOS Transistors With Independent Double GatesDocument3 pagesJunctionless CMOS Transistors With Independent Double GatesBiswajit BeheraNo ratings yet
- Interfacial Reaction and Mechanical Properties of Eutectic Sn-0.7Cu/Ni BGA Solder Joints During Isothermal Long-Term AgingDocument8 pagesInterfacial Reaction and Mechanical Properties of Eutectic Sn-0.7Cu/Ni BGA Solder Joints During Isothermal Long-Term Agingceciliafong51No ratings yet
- Effect of Die Bonding On The Performance of High Power Semiconductor Laser DiodeDocument4 pagesEffect of Die Bonding On The Performance of High Power Semiconductor Laser DiodecjpanchalNo ratings yet
- Bandgap-Brokaw FundamentalsDocument60 pagesBandgap-Brokaw FundamentalsEduardo AvilezNo ratings yet
- Research Proposal MuzamilDocument8 pagesResearch Proposal Muzamilsalman100% (1)
- Conduction at Domain Walls in Insulating PB (ZR Ti) O Thin FilmsDocument13 pagesConduction at Domain Walls in Insulating PB (ZR Ti) O Thin FilmsPN VinodNo ratings yet
- A Generalized Description of The Elastic Properties of NanowiresDocument6 pagesA Generalized Description of The Elastic Properties of NanowiresHugo LivasNo ratings yet
- (2011 Nano Lett) Graphene-Silicon Schottky DiodeDocument5 pages(2011 Nano Lett) Graphene-Silicon Schottky Diodeybgwst5wnrNo ratings yet
- Research ProposalDocument8 pagesResearch ProposalsalmanNo ratings yet
- Characterizations of Cu/Sn-Zn Solder/Ag Interfaces On Photovoltaic Ribbon For Silicon Solar CellsDocument4 pagesCharacterizations of Cu/Sn-Zn Solder/Ag Interfaces On Photovoltaic Ribbon For Silicon Solar CellsKunal JoshiNo ratings yet
- Renewable Energy: E.T. Hu, G.Q. Yue, R.J. Zhang, Y.X. Zheng, L.Y. Chen, S.Y. WangDocument5 pagesRenewable Energy: E.T. Hu, G.Q. Yue, R.J. Zhang, Y.X. Zheng, L.Y. Chen, S.Y. WangAbhimanyu YadavNo ratings yet
- Physics Project On SemiconductorDocument25 pagesPhysics Project On Semiconductorvkanishka1999No ratings yet
- Josephson Junctions - Low-Tc A. ShojiDocument5 pagesJosephson Junctions - Low-Tc A. Shojimosmos1No ratings yet
- Copper Migration Induces Discoloration On A Nipdau SurfaceDocument4 pagesCopper Migration Induces Discoloration On A Nipdau SurfaceJames BelciñaNo ratings yet
- Author's Accepted Manuscript: Energy Storage MaterialsDocument11 pagesAuthor's Accepted Manuscript: Energy Storage MaterialsLu YuNo ratings yet
- Correlation Between Dislocations and Leakage Current of P-N Diodes On A Free-Standing Gan SubstrateDocument5 pagesCorrelation Between Dislocations and Leakage Current of P-N Diodes On A Free-Standing Gan SubstrateAna-Maria DucuNo ratings yet
- Surface Passivation Applicable To Inassb/Gasb Photodiodes For Infrared DetectionDocument6 pagesSurface Passivation Applicable To Inassb/Gasb Photodiodes For Infrared DetectionZohour CherifiNo ratings yet
- Fabrication of Lead Selenide Thin Film Photodiode For Near-Infrared Detection Via O2 Plasma Treatment - Ren Et AlDocument5 pagesFabrication of Lead Selenide Thin Film Photodiode For Near-Infrared Detection Via O2 Plasma Treatment - Ren Et AlGreg McGannNo ratings yet
- Mo Si Multilayers For EUV Lithography by Ion BeamDocument9 pagesMo Si Multilayers For EUV Lithography by Ion BeamEanest HuangNo ratings yet
- Hatakeyama 2011 High Breakdown Voltage GaN PN Junction DiodesDocument3 pagesHatakeyama 2011 High Breakdown Voltage GaN PN Junction Diodesnouman khanNo ratings yet
- Effect of Ni Nanoparticles On Intermetallic Compounds Formation in SAC305 Solder Joint Under High Current DensityDocument5 pagesEffect of Ni Nanoparticles On Intermetallic Compounds Formation in SAC305 Solder Joint Under High Current DensityJimyVillaNo ratings yet
- Fabrication of PN Junction PDFDocument2 pagesFabrication of PN Junction PDFGregg67% (3)
- Development of Zn-Ni-Cd Coatings by Pulse Electrodepostion ProcessDocument12 pagesDevelopment of Zn-Ni-Cd Coatings by Pulse Electrodepostion ProcessjlopezNo ratings yet
- Fabrication: P-N JunctionDocument15 pagesFabrication: P-N JunctionKrishnaveni Subramani SNo ratings yet
- 1361 6528/aa7a54Document10 pages1361 6528/aa7a54hamidNo ratings yet
- Laser Assisted Transfer of Solder Material From A Solid - State Solder Layer For Mask-Less Formation of Micro Solder Depots On Cu-Pillars and ENIG Pad StructuresDocument8 pagesLaser Assisted Transfer of Solder Material From A Solid - State Solder Layer For Mask-Less Formation of Micro Solder Depots On Cu-Pillars and ENIG Pad StructuresYaodong WangNo ratings yet
- LED3Document35 pagesLED3Raja LakshmiNo ratings yet
- Comparing ENIG vs. ENEPIG What's The DifferenceDocument13 pagesComparing ENIG vs. ENEPIG What's The DifferencejackNo ratings yet
- Electronic Circuit I Lecture-1 Part-2 EEE MD Buland Taslim University of ScholarsDocument4 pagesElectronic Circuit I Lecture-1 Part-2 EEE MD Buland Taslim University of ScholarsPakhi SohelNo ratings yet
- Key Hole SizeDocument6 pagesKey Hole Sizeswapnil pandeNo ratings yet
- 1 PN in EquilibriumDocument15 pages1 PN in Equilibriumdeathride007No ratings yet
- International Journal of Electronics and Communications (AEÜ)Document10 pagesInternational Journal of Electronics and Communications (AEÜ)Anonymous 5AmJ13mLkNo ratings yet
- #Substrate Resistance Modeling For Noise Coupling AnalysisDocument6 pages#Substrate Resistance Modeling For Noise Coupling Analysisapi-19668941No ratings yet
- CP System For LPG Underground Tank 1688916092Document5 pagesCP System For LPG Underground Tank 1688916092youplaiNo ratings yet
- P-N Junction Diode Baising and Its VI CharacteristicsDocument12 pagesP-N Junction Diode Baising and Its VI Characteristicsvihan shahNo ratings yet
- A Novel Electrodeposition Process For Plating Zn-Ni-CdDocument10 pagesA Novel Electrodeposition Process For Plating Zn-Ni-CdjlopezNo ratings yet
- Avoiding The Solder VoidDocument7 pagesAvoiding The Solder VoidReza AnvariNo ratings yet
- An Introduction To IGBT Operation: Application NoteDocument5 pagesAn Introduction To IGBT Operation: Application Noterodruren01No ratings yet
- PB3 1Document5 pagesPB3 1womini1025No ratings yet
- Nu-Tie Report 070808mktDocument28 pagesNu-Tie Report 070808mktYam BalaoingNo ratings yet
- SPARK STAR-26HOT-SPOT Non-Current Carrying PartDocument2 pagesSPARK STAR-26HOT-SPOT Non-Current Carrying PartpavanNo ratings yet
- Theory of Dielectric Optical Waveguides 2eFrom EverandTheory of Dielectric Optical Waveguides 2ePaul LiaoNo ratings yet
- Optical Fiber Rotation SensingFrom EverandOptical Fiber Rotation SensingWilliam K. BurnsRating: 3 out of 5 stars3/5 (1)
- Chemistry: Crash Course For JEE Main 2020Document18 pagesChemistry: Crash Course For JEE Main 2020Sanjeeb KumarNo ratings yet
- Movie Piracy in Ethiopian CinemaDocument22 pagesMovie Piracy in Ethiopian CinemaBehailu Shiferaw MihireteNo ratings yet
- Turn Around Coordinator Job DescriptionDocument2 pagesTurn Around Coordinator Job DescriptionMikeNo ratings yet
- Career Level Diagram - V5Document1 pageCareer Level Diagram - V5Shivani RaikwarNo ratings yet
- Technology Management 1Document38 pagesTechnology Management 1Anu NileshNo ratings yet
- Fusion Implementing Offerings Using Functional Setup Manager PDFDocument51 pagesFusion Implementing Offerings Using Functional Setup Manager PDFSrinivasa Rao Asuru0% (1)
- Hole CapacityDocument2 pagesHole CapacityAbdul Hameed OmarNo ratings yet
- FAMOUS PP Past TenseDocument21 pagesFAMOUS PP Past Tenseme me kyawNo ratings yet
- Assembler Pass 2Document5 pagesAssembler Pass 2AnuNo ratings yet
- Laboratory Manual (CIV 210) Engineering Surveying (2018-19) (For Private Circulation Only)Document76 pagesLaboratory Manual (CIV 210) Engineering Surveying (2018-19) (For Private Circulation Only)gyanendraNo ratings yet
- Chapter 2 ProblemsDocument6 pagesChapter 2 ProblemsYour MaterialsNo ratings yet
- Lithuania DalinaDocument16 pagesLithuania DalinaStunt BackNo ratings yet
- Global Geo Reviewer MidtermDocument29 pagesGlobal Geo Reviewer Midtermbusinesslangto5No ratings yet
- On Derridean Différance - UsiefDocument16 pagesOn Derridean Différance - UsiefS JEROME 2070505No ratings yet
- Review1 ScheduleDocument3 pagesReview1 Schedulejayasuryam.ae18No ratings yet
- Hey Friends B TBDocument152 pagesHey Friends B TBTizianoCiro CarrizoNo ratings yet
- Equivalent Fractions Activity PlanDocument6 pagesEquivalent Fractions Activity Planapi-439333272No ratings yet
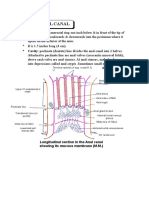
- Anatomy Anal CanalDocument14 pagesAnatomy Anal CanalBela Ronaldoe100% (1)
- The Mantel Colonized Nation Somalia 10 PDFDocument5 pagesThe Mantel Colonized Nation Somalia 10 PDFAhmad AbrahamNo ratings yet
- RSA - Brand - Guidelines - 2019 2Document79 pagesRSA - Brand - Guidelines - 2019 2Gigi's DelightNo ratings yet
- Quick Help For EDI SEZ IntegrationDocument2 pagesQuick Help For EDI SEZ IntegrationsrinivasNo ratings yet
- WEB DESIGN WITH AUSTINE-converted-1Document9 pagesWEB DESIGN WITH AUSTINE-converted-1JayjayNo ratings yet
- Test ScienceDocument2 pagesTest Sciencejam syNo ratings yet
- Tplink Eap110 Qig EngDocument20 pagesTplink Eap110 Qig EngMaciejNo ratings yet
- Technical Bulletin LXL: No. Subject Release DateDocument8 pagesTechnical Bulletin LXL: No. Subject Release DateTrunggana AbdulNo ratings yet
- 3 Carbohydrates' StructureDocument33 pages3 Carbohydrates' StructureDilan TeodoroNo ratings yet
- E0 UoE Unit 7Document16 pagesE0 UoE Unit 7Patrick GutierrezNo ratings yet
- Chemistry Form 4 Daily Lesson Plan - CompressDocument3 pagesChemistry Form 4 Daily Lesson Plan - Compressadila ramlonNo ratings yet
- Strucure Design and Multi - Objective Optimization of A Novel NPR Bumber SystemDocument19 pagesStrucure Design and Multi - Objective Optimization of A Novel NPR Bumber System施元No ratings yet
- Solved Simplex Problems PDFDocument5 pagesSolved Simplex Problems PDFTejasa MishraNo ratings yet