Professional Documents
Culture Documents
Ferrite, also known as α-ferrite (α-Fe) or alpha iron, is a: strain field
Uploaded by
aqhammam0 ratings0% found this document useful (0 votes)
33 views2 pagesFerritic Steel
Original Title
Ferrite
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentFerritic Steel
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
33 views2 pagesFerrite, also known as α-ferrite (α-Fe) or alpha iron, is a: strain field
Uploaded by
aqhammamFerritic Steel
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 2
Ferrite, also known as -ferrite (-Fe) or alpha iron, is a materials science term for pure iron, with
a body-centered cubic B.C.C crystal structure. It is this crystalline structure which
gives steel and cast iron their magnetic properties, and is the classic example of
a ferromagnetic material.[1]
It has a strength of 280 N/mm2[citation needed] and a hardness of approximately 80 Brinell.[2]
Mild steel (carbon steel with up to about 0.2 wt% C) consist mostly of ferrite, with increasing
amounts of pearlite (a fine lamellar structure of ferrite and cementite) as the carbon content is
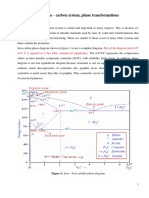
increased. Since bainite (shown as ledeburite on the diagram at the bottom of this page) and pearlite
each have ferrite as a component, any iron-carbon alloy will contain some amount of ferrite if it is
allowed to reach equilibrium at room temperature. The exact amount of ferrite will depend on the
cooling processes the iron-carbon alloy undergoes as it cools from liquid state.
In pure iron, ferrite is stable below 910 C (1,670 F). Above this temperature the face-centred
cubic form of iron,austenite (gamma-iron) is stable. Above 1,390 C (2,530 F), up to the melting
point at 1,539 C (2,802 F), the body-centred cubic crystal structure is again the more stable form
of delta-ferrite (-Fe). Ferrite above the critical temperature A2 (Curie temperature) of 771 C
(1,044 K; 1,420 F), where it is paramagnetic rather than ferromagnetic, is beta ferrite or beta iron (Fe). The term beta iron is seldom used because it is crystallographically identical to, and its phase
field contiguous with, -Fe.
Only a very small amount of carbon can be dissolved in ferrite; [3] the maximum solubility is about
0.02 wt% at 723 C (1,333 F) and 0.005% carbon at 0 C (32 F).[4] This is because carbon
dissolves in iron interstitially, with the carbon atoms being about twice the diameter of the interstitial
"holes", so that each carbon atom is surrounded by a strong local strain field. Hence the enthalpy of
mixing is positive (unfavourable), but the contribution of entropy to the free
energy of solution stabilises the structure for low carbon content. 723 C (1,333 F) also is the
minimum temperature at which iron-carbon austenite (0.8 wt% C) is stable; at this temperature there
is a eutectoid reaction between ferrite, austenite and cementite.
Molar volume vs. pressure for -ferrite at room temperature.
Because of its significance for planetary cores, the physical properties of iron at high pressures and
temperatures have also been studied extensively. -ferrite, which is the form of iron that is stable
under standard conditions, can be subjected to pressures up to ca. 15 GPa before transforming into
a high-pressure form termed -iron, which crystallizes in a hexagonal close-packed (hcp) structure.
You might also like
- The Working of Steel: Annealing, Heat Treating and Hardening of Carbon and Alloy SteelFrom EverandThe Working of Steel: Annealing, Heat Treating and Hardening of Carbon and Alloy SteelNo ratings yet
- Iron Carbon Equilibrium DiagramDocument4 pagesIron Carbon Equilibrium DiagramParameshwari PrabakarNo ratings yet
- Allotropes of IronDocument3 pagesAllotropes of IronKarthik RangarajNo ratings yet
- The Iron-Carbon Phase DiagramDocument16 pagesThe Iron-Carbon Phase DiagramMeena SivasubramanianNo ratings yet
- IRON CARBON Equilibrium Diagram BME 3Document4 pagesIRON CARBON Equilibrium Diagram BME 3sahitya karaheNo ratings yet
- Allotropes of IronDocument5 pagesAllotropes of IronShamsul RamliNo ratings yet
- The Iron-Carbon Equilibrium Diagram: AbstractDocument4 pagesThe Iron-Carbon Equilibrium Diagram: Abstractleodavid87No ratings yet
- Fe-C Phase Diagram ExplainedDocument5 pagesFe-C Phase Diagram ExplainednareshNo ratings yet
- The Iron-Carbon Equilibrium Diagram: AbstractDocument4 pagesThe Iron-Carbon Equilibrium Diagram: AbstractRama Krishna Reddy DonthireddyNo ratings yet
- Phase Diagram of Fe-Fe3CDocument25 pagesPhase Diagram of Fe-Fe3CIram MustaviNo ratings yet
- Iron Carbon DiagramDocument23 pagesIron Carbon DiagramdeepakNo ratings yet
- Metallury of SteelsDocument10 pagesMetallury of SteelsDalitso MwanzaNo ratings yet
- University of Babylon, College of Engineering, Engineering Metallurgy, Maithem H-RasheedDocument13 pagesUniversity of Babylon, College of Engineering, Engineering Metallurgy, Maithem H-RasheedAris BulaongNo ratings yet
- Cast Steel: The Iron-Carbon Equilibrium Diagram: AbstractDocument5 pagesCast Steel: The Iron-Carbon Equilibrium Diagram: Abstractchacha4500No ratings yet
- Ironiron CarbideequilibriumphasediagramDocument39 pagesIroniron CarbideequilibriumphasediagramSheikh UMARNo ratings yet
- Iron - Carbon SystemDocument21 pagesIron - Carbon SystemYavana KeerthiNo ratings yet
- Lesson 5 - Fe-C Diagram - Rev. 0Document11 pagesLesson 5 - Fe-C Diagram - Rev. 0Arga SetyaNo ratings yet
- Steels: Ii Beng (Hons) Mech Eng (Well Eng) Metallurgy & Manufacturing ScienceDocument39 pagesSteels: Ii Beng (Hons) Mech Eng (Well Eng) Metallurgy & Manufacturing ScienceKareem YasserNo ratings yet
- Introduction-Iron Carbon Phase DiagramDocument31 pagesIntroduction-Iron Carbon Phase DiagramTHE BBEASTNo ratings yet
- Iron Carbon DiagramDocument10 pagesIron Carbon DiagramsivakumarNo ratings yet
- Iron Carbon Equillibrium Diagram GandhidhamDocument22 pagesIron Carbon Equillibrium Diagram Gandhidhamcal2_uniNo ratings yet
- Iron Forms 2Document6 pagesIron Forms 2muralisrikanthNo ratings yet
- 3 Iron Carbon DiaDocument21 pages3 Iron Carbon DiaChhavi SharmaNo ratings yet
- Ch-27.5 Iron Carbon Equilibrium DiagramDocument52 pagesCh-27.5 Iron Carbon Equilibrium DiagramManojNo ratings yet
- MSM GTU Study Material E-Notes Unit-5 23112020052908AMDocument14 pagesMSM GTU Study Material E-Notes Unit-5 23112020052908AMVijayNo ratings yet
- Industrial Materials: Instructed By: Dr. Sajid ZaidiDocument13 pagesIndustrial Materials: Instructed By: Dr. Sajid ZaidiarulmuruguNo ratings yet
- Allotropes of IronDocument5 pagesAllotropes of IronVysakh VasudevanNo ratings yet
- Iron - Carbon Phase Diagram: Sandeep Nair CB - EN.P2MFG15018Document30 pagesIron - Carbon Phase Diagram: Sandeep Nair CB - EN.P2MFG15018prasenjitsayantan100% (1)
- Engineering Metallurgy: Misan University-College of EngineeringDocument26 pagesEngineering Metallurgy: Misan University-College of Engineeringbone manNo ratings yet
- The Microstructural Nature of Carbon Steels Phase DiagramDocument4 pagesThe Microstructural Nature of Carbon Steels Phase Diagramamitkharb111195No ratings yet
- Fe-C DiagramDocument7 pagesFe-C DiagramYaser Mohamed AbasNo ratings yet
- M. Tech. (FFT) Technology of Ferrous Casting Phase DiagramDocument7 pagesM. Tech. (FFT) Technology of Ferrous Casting Phase DiagramRajulapati Sunil KumarNo ratings yet
- Ch-27.3 Iron Carbon Equilibrium DiagramDocument58 pagesCh-27.3 Iron Carbon Equilibrium DiagramasjfgauojfgfNo ratings yet
- AUSTENITEDocument4 pagesAUSTENITESaba ShaikhNo ratings yet
- Iron Carbon Note 1 2023Document23 pagesIron Carbon Note 1 2023gerrard samuelNo ratings yet
- Metallurgy AssignmentDocument9 pagesMetallurgy Assignmentvishnupriya somaganiNo ratings yet
- Metallurgy AssignmentDocument9 pagesMetallurgy Assignmentvishnupriya somaganiNo ratings yet
- Iron-Carbide Phase Diagram AnalysisDocument26 pagesIron-Carbide Phase Diagram AnalysisHiral HiraniNo ratings yet
- Iron Carbon Phase DiagramDocument4 pagesIron Carbon Phase DiagramMizanur RahmanNo ratings yet
- Iron - Iron Carbide Iron Iron Carbide Phase Diagram: Today's TopicsDocument11 pagesIron - Iron Carbide Iron Iron Carbide Phase Diagram: Today's TopicsembargioNo ratings yet
- Plain Iron Carbon SteelsDocument5 pagesPlain Iron Carbon Steelsروشان فاطمة روشانNo ratings yet
- Review of steel microstructures and phasesDocument3 pagesReview of steel microstructures and phasesVajid MadathilNo ratings yet
- What Is PearliteDocument4 pagesWhat Is Pearliteardy cornettoNo ratings yet
- Iron-Carbon Phase DiagramDocument30 pagesIron-Carbon Phase Diagramjunaid hassanNo ratings yet
- Engineering Material II Short NoteDocument17 pagesEngineering Material II Short NotewondimuNo ratings yet
- The IronCarbide DiagramDocument11 pagesThe IronCarbide DiagramshajjikhalidNo ratings yet
- Iron-Carbide DiagramDocument6 pagesIron-Carbide DiagramAbhijit GhanwatNo ratings yet
- Engineering Alloys (Ferrous)Document103 pagesEngineering Alloys (Ferrous)Sukhwinder Singh GillNo ratings yet
- FMP 221 Lecture 4Document22 pagesFMP 221 Lecture 4SarojKumarSinghNo ratings yet
- Microstructure Differences Between Base Metal, Weld Metal and HAZDocument3 pagesMicrostructure Differences Between Base Metal, Weld Metal and HAZShaikhan Nadzemi100% (1)
- Definitions and Related Materials: Stahliją or Stakhlijan (Made of Steel), Stahliją (Standing Firm)Document5 pagesDefinitions and Related Materials: Stahliją or Stakhlijan (Made of Steel), Stahliją (Standing Firm)kingNo ratings yet
- Iron-Carbon Phase Diagram: By: Awad Elaraby ID:052022009Document33 pagesIron-Carbon Phase Diagram: By: Awad Elaraby ID:052022009Mahmoud RefaatNo ratings yet
- Iron Carbon DiagramDocument8 pagesIron Carbon Diagramashok pradhanNo ratings yet
- Lec 7 Fe C DiagramDocument45 pagesLec 7 Fe C DiagramAdnan MehmoodNo ratings yet
- Phase Diagrams:: The Iron-Iron Carbide (Fe-Fe3C) Diagram or Iron-Carbon (Fe-C) Equilibrium DiagramDocument46 pagesPhase Diagrams:: The Iron-Iron Carbide (Fe-Fe3C) Diagram or Iron-Carbon (Fe-C) Equilibrium DiagramUsman FarooqNo ratings yet
- Austenite: Iron-carbon phase diagram, showing the conditions under which austenite (γ) is stable in carbon steelDocument4 pagesAustenite: Iron-carbon phase diagram, showing the conditions under which austenite (γ) is stable in carbon steelVysakh VasudevanNo ratings yet
- Austenite Perlite TransformationDocument1 pageAustenite Perlite TransformationkapsarcNo ratings yet
- Metallurgy of Carbon SteelDocument5 pagesMetallurgy of Carbon SteelMadhavan SoundararajanNo ratings yet
- Lecture 9 - Ferrous AlloysDocument31 pagesLecture 9 - Ferrous Alloysmahmoud foudaNo ratings yet
- Gate HydraulicDocument2 pagesGate HydraulicaqhammamNo ratings yet
- Calculating Recommended Hose Diameter in A Hydraulic Circuit - FlowfitDocument3 pagesCalculating Recommended Hose Diameter in A Hydraulic Circuit - FlowfitaqhammamNo ratings yet
- Compressed Air FlowmeterDocument2 pagesCompressed Air FlowmeteraqhammamNo ratings yet
- Rate Analysis: 1 199.5 PKR 1 $ 175.95 PKR NBP FX Rate Sheet 15.2.2022 NBP FX Rate Sheet 15.2.2023Document1 pageRate Analysis: 1 199.5 PKR 1 $ 175.95 PKR NBP FX Rate Sheet 15.2.2022 NBP FX Rate Sheet 15.2.2023aqhammamNo ratings yet
- Calculating Recommended Hose Diameter in A Hydraulic Circuit - FlowfitDocument3 pagesCalculating Recommended Hose Diameter in A Hydraulic Circuit - FlowfitaqhammamNo ratings yet
- World Wide Metric BlogDocument2 pagesWorld Wide Metric BlogaqhammamNo ratings yet
- Selecting The Correct Hydraulic Oil - Hydraulics & PneumaticsDocument5 pagesSelecting The Correct Hydraulic Oil - Hydraulics & PneumaticsaqhammamNo ratings yet
- Gate HyDocument2 pagesGate HyaqhammamNo ratings yet
- How Do You Know If You're Using The Right Hydraulic Oil - Machinery LubricationDocument4 pagesHow Do You Know If You're Using The Right Hydraulic Oil - Machinery LubricationaqhammamNo ratings yet
- RG Group ArticleDocument4 pagesRG Group ArticleaqhammamNo ratings yet
- Viscosity of Hydraulic OilDocument2 pagesViscosity of Hydraulic OilaqhammamNo ratings yet
- Safety 2Document1 pageSafety 2aqhammamNo ratings yet
- Stamp: Before You ItDocument6 pagesStamp: Before You ItaqhammamNo ratings yet
- How Do I Pick The Right Hydraulic Hose 2Document2 pagesHow Do I Pick The Right Hydraulic Hose 2aqhammamNo ratings yet
- Sizing Tubes To Maximize Hydraulic Efficiency - Machine DesignDocument7 pagesSizing Tubes To Maximize Hydraulic Efficiency - Machine DesignaqhammamNo ratings yet
- Tips On Selection of Plumbing Size For Hydraulic Systems - Womack Machine Supply CompanyDocument3 pagesTips On Selection of Plumbing Size For Hydraulic Systems - Womack Machine Supply CompanyaqhammamNo ratings yet
- HEALTHDocument1 pageHEALTHaqhammamNo ratings yet
- Why You Shouldn't Overlook Hydraulic Pipe SizeDocument4 pagesWhy You Shouldn't Overlook Hydraulic Pipe SizeaqhammamNo ratings yet
- Rate Analysis for Fabrication and Installation WorkDocument8 pagesRate Analysis for Fabrication and Installation WorkaqhammamNo ratings yet
- ENVIRONMENTDocument1 pageENVIRONMENTaqhammamNo ratings yet
- Code of ConductDocument1 pageCode of ConductaqhammamNo ratings yet
- Anti-Corruption Anti-Corruption Policy PolicyDocument1 pageAnti-Corruption Anti-Corruption Policy PolicyaqhammamNo ratings yet
- Policy On Corporate Social Responsibility Social Responsibility (CSR) (CSR)Document1 pagePolicy On Corporate Social Responsibility Social Responsibility (CSR) (CSR)aqhammamNo ratings yet
- Rate Analysis for Fabrication and Installation WorkDocument8 pagesRate Analysis for Fabrication and Installation WorkaqhammamNo ratings yet
- Rate Analysis of Super StructureDocument8 pagesRate Analysis of Super StructureaqhammamNo ratings yet
- Rate Analysis of Super StructureDocument8 pagesRate Analysis of Super StructureaqhammamNo ratings yet
- Chain PulleyDocument3 pagesChain PulleyaqhammamNo ratings yet
- Materials Notes 15 Rolling - Forging PDFDocument6 pagesMaterials Notes 15 Rolling - Forging PDFaqhammamNo ratings yet
- SBP Rate Sheet 09 Nov 2018Document1 pageSBP Rate Sheet 09 Nov 2018aqhammamNo ratings yet
- Moveable Span Bridge Study Volume 2 Bascule and Swing Span Bridges Part 2 PDFDocument54 pagesMoveable Span Bridge Study Volume 2 Bascule and Swing Span Bridges Part 2 PDFaqhammam100% (1)
- Mil B 49430BDocument36 pagesMil B 49430Bparam.vennelaNo ratings yet
- E Requisition SystemDocument8 pagesE Requisition SystemWaNi AbidNo ratings yet
- History of Computer AnimationDocument39 pagesHistory of Computer AnimationRanzelle UrsalNo ratings yet
- Internal Controls and Risk Management: Learning ObjectivesDocument24 pagesInternal Controls and Risk Management: Learning ObjectivesRamil SagubanNo ratings yet
- Develop Network Monitoring System IP Subnet CalculatorDocument23 pagesDevelop Network Monitoring System IP Subnet CalculatorabiramanNo ratings yet
- Margin Philosophy For Science Assessment Studies: EstecDocument11 pagesMargin Philosophy For Science Assessment Studies: EstecFeyippNo ratings yet
- Science-6 - Q4 - W8-DLL - Mar 10Document2 pagesScience-6 - Q4 - W8-DLL - Mar 10cristina quiambaoNo ratings yet
- Usage Instructions - Hengro PoolProtect 1 - 5Document3 pagesUsage Instructions - Hengro PoolProtect 1 - 5Brian VisserNo ratings yet
- 1 s2.0 S1747938X21000142 MainDocument33 pages1 s2.0 S1747938X21000142 MainAzmil XinanNo ratings yet
- Documentation For UStarDocument26 pagesDocumentation For UStarthunder77No ratings yet
- Information HandoutsDocument6 pagesInformation HandoutsPooja Marwadkar TupcheNo ratings yet
- C172M QRH (VH-JZJ) v1.1Document49 pagesC172M QRH (VH-JZJ) v1.1alphaNo ratings yet
- How To Approach To Case Study Type Questions and MCQsDocument4 pagesHow To Approach To Case Study Type Questions and MCQsKushang ShahNo ratings yet
- University Reserch Worksheet PDFDocument7 pagesUniversity Reserch Worksheet PDFapi-410567922No ratings yet
- Be3000 Manual 2016Document77 pagesBe3000 Manual 2016Itzel JuárezNo ratings yet
- PMMAEE Application FormDocument2 pagesPMMAEE Application Formshiero501100% (15)
- Amber ToolsDocument309 pagesAmber ToolshkmydreamsNo ratings yet
- wt-and-fds-practical-slipsDocument32 pageswt-and-fds-practical-slipsRohidas SapkalNo ratings yet
- Xiaomi Mi Drone 4K User Manual GuideDocument47 pagesXiaomi Mi Drone 4K User Manual GuideΜιχάλης ΛαχανάςNo ratings yet
- Lecture 6-Design For ShearDocument14 pagesLecture 6-Design For ShearMarkos DanielNo ratings yet
- Technology and Livelihood Education: Agri - Fishery Arts (Agricultural Crops Production) Marketing Agricultural ProductsDocument14 pagesTechnology and Livelihood Education: Agri - Fishery Arts (Agricultural Crops Production) Marketing Agricultural Productslana del rey100% (1)
- NACE CIP Part II - (6) Coatings For Industry - (Qs - As)Document23 pagesNACE CIP Part II - (6) Coatings For Industry - (Qs - As)Almagesto QuenayaNo ratings yet
- 3.1-Pile Design Calculation For Boundary (p1 To p50)Document24 pages3.1-Pile Design Calculation For Boundary (p1 To p50)layaljamal2No ratings yet
- Tomas Del Rosario College: Department: EDUCATIONDocument12 pagesTomas Del Rosario College: Department: EDUCATIONveehneeNo ratings yet
- HRM Assignment Final - Case StudyDocument7 pagesHRM Assignment Final - Case StudyPulkit_Bansal_2818100% (3)
- Plenaristas León 2022xDocument6 pagesPlenaristas León 2022xGloria MontielNo ratings yet
- 2VAA001695 en S Control NTCS04 Controller Station Termination UnitDocument43 pages2VAA001695 en S Control NTCS04 Controller Station Termination UnitanbarasanNo ratings yet
- REFLEKSI KASUS PLASENTADocument48 pagesREFLEKSI KASUS PLASENTAImelda AritonangNo ratings yet
- CH 11 & CH 12 John R. Schermerhorn - Management-Wiley (2020)Document16 pagesCH 11 & CH 12 John R. Schermerhorn - Management-Wiley (2020)Muhammad Fariz IbrahimNo ratings yet
- Hyd Schematic 2658487205 - 005Document3 pagesHyd Schematic 2658487205 - 005Angelo Solorzano100% (2)
- Quantum Physics: What Everyone Needs to KnowFrom EverandQuantum Physics: What Everyone Needs to KnowRating: 4.5 out of 5 stars4.5/5 (48)
- Quantum Spirituality: Science, Gnostic Mysticism, and Connecting with Source ConsciousnessFrom EverandQuantum Spirituality: Science, Gnostic Mysticism, and Connecting with Source ConsciousnessRating: 4 out of 5 stars4/5 (6)
- Quantum Physics for Beginners Who Flunked Math And Science: Quantum Mechanics And Physics Made Easy Guide In Plain Simple EnglishFrom EverandQuantum Physics for Beginners Who Flunked Math And Science: Quantum Mechanics And Physics Made Easy Guide In Plain Simple EnglishRating: 4.5 out of 5 stars4.5/5 (18)
- Too Big for a Single Mind: How the Greatest Generation of Physicists Uncovered the Quantum WorldFrom EverandToo Big for a Single Mind: How the Greatest Generation of Physicists Uncovered the Quantum WorldRating: 4.5 out of 5 stars4.5/5 (8)
- Summary and Interpretation of Reality TransurfingFrom EverandSummary and Interpretation of Reality TransurfingRating: 5 out of 5 stars5/5 (5)
- The Tao of Physics: An Exploration of the Parallels between Modern Physics and Eastern MysticismFrom EverandThe Tao of Physics: An Exploration of the Parallels between Modern Physics and Eastern MysticismRating: 4 out of 5 stars4/5 (500)
- Midnight in Chernobyl: The Story of the World's Greatest Nuclear DisasterFrom EverandMidnight in Chernobyl: The Story of the World's Greatest Nuclear DisasterRating: 4.5 out of 5 stars4.5/5 (409)
- A Brief History of Time: From the Big Bang to Black HolesFrom EverandA Brief History of Time: From the Big Bang to Black HolesRating: 4 out of 5 stars4/5 (2193)
- The Physics of God: How the Deepest Theories of Science Explain Religion and How the Deepest Truths of Religion Explain ScienceFrom EverandThe Physics of God: How the Deepest Theories of Science Explain Religion and How the Deepest Truths of Religion Explain ScienceRating: 4.5 out of 5 stars4.5/5 (23)
- Packing for Mars: The Curious Science of Life in the VoidFrom EverandPacking for Mars: The Curious Science of Life in the VoidRating: 4 out of 5 stars4/5 (1395)
- The End of Everything: (Astrophysically Speaking)From EverandThe End of Everything: (Astrophysically Speaking)Rating: 4.5 out of 5 stars4.5/5 (155)
- Bedeviled: A Shadow History of Demons in ScienceFrom EverandBedeviled: A Shadow History of Demons in ScienceRating: 5 out of 5 stars5/5 (5)
- Lost in Math: How Beauty Leads Physics AstrayFrom EverandLost in Math: How Beauty Leads Physics AstrayRating: 4.5 out of 5 stars4.5/5 (125)
- The Magick of Physics: Uncovering the Fantastical Phenomena in Everyday LifeFrom EverandThe Magick of Physics: Uncovering the Fantastical Phenomena in Everyday LifeNo ratings yet
- Strange Angel: The Otherworldly Life of Rocket Scientist John Whiteside ParsonsFrom EverandStrange Angel: The Otherworldly Life of Rocket Scientist John Whiteside ParsonsRating: 4 out of 5 stars4/5 (94)
- The Holographic Universe: The Revolutionary Theory of RealityFrom EverandThe Holographic Universe: The Revolutionary Theory of RealityRating: 4.5 out of 5 stars4.5/5 (75)
- What is Life?: With Mind and Matter and Autobiographical SketchesFrom EverandWhat is Life?: With Mind and Matter and Autobiographical SketchesRating: 4 out of 5 stars4/5 (139)
- The Beginning of Infinity: Explanations That Transform the WorldFrom EverandThe Beginning of Infinity: Explanations That Transform the WorldRating: 5 out of 5 stars5/5 (60)
- Starry Messenger: Cosmic Perspectives on CivilizationFrom EverandStarry Messenger: Cosmic Perspectives on CivilizationRating: 4.5 out of 5 stars4.5/5 (158)
- Black Holes: The Key to Understanding the UniverseFrom EverandBlack Holes: The Key to Understanding the UniverseRating: 4.5 out of 5 stars4.5/5 (13)
- The Sounds of Life: How Digital Technology Is Bringing Us Closer to the Worlds of Animals and PlantsFrom EverandThe Sounds of Life: How Digital Technology Is Bringing Us Closer to the Worlds of Animals and PlantsRating: 5 out of 5 stars5/5 (5)
- The Power of Eight: Harnessing the Miraculous Energies of a Small Group to Heal Others, Your Life, and the WorldFrom EverandThe Power of Eight: Harnessing the Miraculous Energies of a Small Group to Heal Others, Your Life, and the WorldRating: 4.5 out of 5 stars4.5/5 (53)