Professional Documents
Culture Documents
An Old Friend Revisited: Chloramphenicol Optic Neuropathy: Case Report
Uploaded by
ckeshavaOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
An Old Friend Revisited: Chloramphenicol Optic Neuropathy: Case Report
Uploaded by
ckeshavaCopyright:
Available Formats
CASE REPORT
An old friend revisited:
chloramphenicol optic
neuropathy
SH Wong1,2
F Silva2 JF Acheson1,2 GT Plant1,2
Department of Neuro-ophthalmology, Moorfields Eye Hospital, London, UK
Department of Neuro-ophthalmology, National Hospital for Neurology and Neurosurgery, London, UK
Correspondence to: Dr Sui Wong, Department of Neuro-ophthalmology, Moorfields Eye Hospital, 162 City Road, London,
EC1V 2PD, UK. Email: suiwong@doctors.org.uk
DECLARATIONS
Competing interests
None declared
With increasing antibiotic resistance, oral chloramphenicol may be utilized more frequently;
below we highlight the risk of toxic optic
neuropathy.
Funding
None declared
Case report
Ethical approval
Written consent was
received from the
patient
Guarantor
SHW
Contributorship
All authors were
involved in the
conception of the
case report and in
its editing
Acknowledgements
We thank the patient
for consent to
present her case.
This case was
presented at the
Association of
British Neurologists
annual meeting,
June 2012
Reviewer
Ali Yagan
A 66-year-old woman presented on October 2011
with a 10-day history of painless bilateral visual
loss of subacute onset (over 48 h). She also
described paraesthesiae of the limbs for a similar
duration.
She has rheumatoid arthritis, treated with
methotrexate (since 2002) and etanercept (2003
2009), and she required bilateral knee replacements in 2009. Her right knee replacement was
revised and underwent six washouts for persistent
infection. Previous antibiotics used for her knee
infection included ceftriaxone, fusidic acid, amoxicillin, vancomycin and daptomycin. Chloramphenicol 4 g daily was started 14 weeks before
the occurrence of visual loss.
Chloramphenicol had been discontinued five
days before presentation. Her other medications
included methotrexate, folic acid, tramadol, paracetamol, aspirin and omeprazole. She smokes 10
cigarettes daily and does not drink alcohol. She
has a varied balanced diet. There is no relevant
family history.
On examination at 10 days after onset of visual
loss, she had a best corrected visual acuity of 3/24
(Snellen) bilaterally and centrocaecal scotomas on
examination by confrontation with a red hat pin.
None of the Ishihara colour plates could be read,
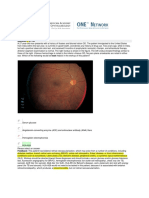
not even the control plate. Both her optic discs
were hyperaemic (see Figure 1). The rest of her
cranial nerves and neurological examination of
limbs were normal, except for bilateral L5 dermatomal loss to pinprick and abnormal proprioception at the great toe. Both temporal arteries were
normal to palpation.
Investigations showed normochromic normocytic anaemia (haemoglobin 10.9 g/dL, mean corpuscular volume 94 fL). C-reactive protein was
elevated at 54.7 mg/L (normal <5). The rest of
blood tests were unremarkable including vitamin
B12, folate, methylmalonic acid, homocysteine,
autoantibodies (antinuclear antibody, extractable
nuclear antigen and antineutrophil cytoplasmic
antibody) and a genetic screen for Leber Hereditary Optic Neuropathy mutations.
Magnetic resonance imaging (MRI) of brain
and orbits was normal and MRI spine showed
mild degenerative changes at cervical spine only.
Cerebrospinal fluid examination was normal.
A visual evoked potential was performed after
recovery and this was normal, as were nerve
conduction studies.
A diagnosis of toxic optic neuropathy secondary to chloramphenicol was suspected. As stated
above, the chloramphenicol had been stopped
five days before presentation to us, i.e. on day 6
of visual symptoms. Four weeks after stopping
chloramphenicol, vision improved to acuities of
6/5 (right), 6/6 (left) and normal visual fields,
but reading of the Ishihara colour plates remained
impaired at 8/13 bilaterally.
The patient elected to have an above knee
amputation of her right leg, and is now mobilizing
with a prosthetic limb and rehabilitation. At the
onset of visual symptoms she developed paraesthesiae of limbs. Although the paraesthesiae
improved, she is still symptomatic with this and
the sensory loss in the left foot is impairing her
prosthesis use.
J R Soc Med Sh Rep 2013;4:20. DOI 10.1177/2042533313476692
Journal of the Royal Society of Medicine Short Reports
Figure 1
Bilateral hyperaemic optic discs seen in our patient with chloramphenicol-associated toxic optic
neuropathy
Discussion
Chloramphenicol oral therapy was commonly
used until the late 1980s, but with the publicized
idiosyncratic reaction of bone marrow suppression1 and the availability of newer antibiotics, its
use was slowly phased out, at least in the developed world.2 However, with the increasing emergence of antibiotic resistance, older generations
of antibiotics may potentially be used more frequently, as was the case in our patient. The previously reported adverse reactions to these older
generation antibiotics may therefore be less
familiar to the current generation of physicians.
Toxic optic neuropathy secondary to chloramphenicol was first described in 1950. Approximately 40 cases of chloramphenicol optic
neuropathy were reported from 1950 to 1988, but
for 23 years since then only two cases were
reported,3 7 a trend that may reflect the reduction
in chloramphenicol use.
The bone marrow suppression caused by chloramphenicol has long been recognized by physicians,1 leading to a severe idiosyncratic adverse
reaction even with brief courses of treatment. By
contrast, chloramphenicol toxic optic neuropathy
is associated with prolonged use (more than
6 weeks) and high cumulative dose (>100 g).8
Knowledge of these risk factors, by avoiding prolonged courses or high doses, can potentially
avoid this adverse reaction.
J R Soc Med Sh Rep 2013;4:20. DOI 10.1177/2042533313476692
The visual loss in chloramphenicol toxic optic
neuropathy may be sudden or subacute, and
painful or painless. There may often be limb
paraesthesiae preceding the visual symptoms.4
The typical ocular signs are bilateral optic disc
swelling, retinal vessel tortuosity and retinal
haemorrhages; our patients optic discs were certainly hyperaemic, and possibly slightly swollen
(see Figure 1). However, the fundi may also be
normal. A centrocaecal scotoma is typically seen,
and is pathognomonic for toxic/nutritional optic
neuropathies. The differential diagnoses to be
considered were nutritional optic neuropathy
and Leber Hereditary Optic Neuropathy, both
of which were excluded in our patient. Hypotheses suggested include mitochondrial inhibition and vitamin B metabolism inhibition by
chloramphenicol.4,9
The treatment is to stop chloramphenicol,
which often leads to good, or even full, recovery
of vision.3,4,6 There is anecdotally reported use of
high-dose vitamin B ( pyridoxine and cyanocobalamin) but the evidence for this is not robust.8,10
Our patient recovered simply with stopping the
offending drug.
Conclusion
We describe a case of toxic optic neuropathy
secondary to chloramphenicol. Chloramphenicol
Chloramphenicol optic neuropathy
optic neuropathy causes subacute visual loss with
centrocaecal scotoma. The risk factors are prolonged treatment (>6 weeks) and a cumulative
dose of >100 g. The management is to stop the
drug. This case highlights the potential pitfalls
resulting from the use of older generation antibiotics and lack of familiarity with their adverse
effects. The problem is likely to become increasingly pertinent as antibiotic resistance increases.
References
1
2
3
Best WR. Chloramphenicol-associated blood dyscrasias.
JAMA 1967;201:181 8
Gump DW. Chloramphenicol: a 1981 view. Arch Intern Med
Am Med Assoc 1981;141:573
Godel V, Nemet P, Lazar M. Chloramphenicol optic
neuropathy. Arch Ophthalmol 1980;98:1417 21
10
Harley RD, Huang NN, Macri CH, Green WR. Optic
neuritis and optic atrophy following chloramphenicol in
cystic fibrosis patients. Trans Am Acad Ophthalmol
Otolaryngol 1970;74:1011 31
Joy R, Scalettar R, Sodee D. Optic and peripheral neuritis.
Probable effect of prolonged chloramphenicol therapy.
JAMA 1960;173:1731 4
Chang N, Giles C, Gregg R. Optic neuritis and
chloramphenicol. Archives of pediatrics and adolescent
medicine. Am Med Assoc 1966;112:46
Fung AT, Hudson B, Billson FA. Chloramphenicol not so
innocuous: a case of optic neuritis. BMJ Case Rep 2011
Mar 3; pii: bcr1020103434
Huang NN, Harley RD, Promadhattavedi V, Sproul A.
Visual disturbances in cystic fibrosis following
chloramphenicol administration. J Pediatrics 1966;68:32 44
Carelli V, Ross-Cisneros FN, Sadun AA. Optic nerve
degeneration and mitochondrial dysfunction: genetic and
acquired optic neuropathies. Neurochem Int 2002;40:573
Cocke JG. Chloramphenicol optic neuritis. Apparent
protective effects of very high daily doses of pyridoxine
and cyanocobalamin. Am J Dis Child 1967;114:424 6
# 2013 The Author(s)
This is an open-access article distributed under the terms of the Creative Commons Non-commercial Attribution
License (http://creativecommons.org/licenses/by-nc/2.0/), which permits non-commercial use, distribution and
reproduction in any medium, provided the original work is properly cited.
J R Soc Med Sh Rep 2013;4:20. DOI 10.1177/2042533313476692
You might also like
- Nihms 478821Document13 pagesNihms 478821AmeldaNo ratings yet
- Nihms 478821Document13 pagesNihms 478821AmeldaNo ratings yet
- Respiratory Critical CareDocument323 pagesRespiratory Critical CarePriyadarshini Varadaraj100% (6)
- Recumbant DogDocument2 pagesRecumbant DogDanielle Lynn PeckNo ratings yet
- Leprosy in OrthopedicsDocument91 pagesLeprosy in OrthopedicsAnkit Agur67% (3)
- Review of Optic Neuritis Treatment TrialDocument5 pagesReview of Optic Neuritis Treatment TrialsatrianiNo ratings yet
- Optic Neuropathy Without Retinopathy in AIDS and Cytomegalovirus InfectionDocument6 pagesOptic Neuropathy Without Retinopathy in AIDS and Cytomegalovirus InfectionJoyeeta DasNo ratings yet
- Miller Fisher Syndrome Mimicking Ocular Myasthenia GravisDocument10 pagesMiller Fisher Syndrome Mimicking Ocular Myasthenia GravisMonica Lauretta Sembiring IINo ratings yet
- Herpes Zoster OphthalmicusDocument12 pagesHerpes Zoster OphthalmicusGio Vano NaihonamNo ratings yet
- Chloroquine/Hydroxychloroquine Toxicity: Differential Diagnoses & Workup Treatment & Medication Follow-Up MultimediaDocument7 pagesChloroquine/Hydroxychloroquine Toxicity: Differential Diagnoses & Workup Treatment & Medication Follow-Up MultimediaVeeranna LokapurNo ratings yet
- Orbital Apex Syndrome With Encephalitis A Rare and Serious Complication of Herpes Zoster Opthalmicus - Ludwig Melino Tjokrovonco 2Document12 pagesOrbital Apex Syndrome With Encephalitis A Rare and Serious Complication of Herpes Zoster Opthalmicus - Ludwig Melino Tjokrovonco 2Handi KrisnaniNo ratings yet
- Tacrolimus Optic NeuropathyDocument7 pagesTacrolimus Optic NeuropathyVlady BordaNo ratings yet
- Commentary: The Optic Neuritis Treatment TrialDocument2 pagesCommentary: The Optic Neuritis Treatment TrialEmmanuel Zótico Orellana JiménezNo ratings yet
- Topical TreatmentDocument7 pagesTopical TreatmentranpssNo ratings yet
- 01 WNL 0000042787 51461 d1Document2 pages01 WNL 0000042787 51461 d1veronikiNo ratings yet
- Coats Disease: Classification and Treatment: by Rishav Kansal, Bs Kiran Turaka, MD and Carol L. Shields, MDDocument3 pagesCoats Disease: Classification and Treatment: by Rishav Kansal, Bs Kiran Turaka, MD and Carol L. Shields, MDAnggie Imaniah SitompulNo ratings yet
- Systemic Lupus ErythematosusDocument5 pagesSystemic Lupus ErythematosusadibahtiarNo ratings yet
- HLA B-27 Associated Anterior Uveitis: American Academy of Optometry: Case Report #1Document22 pagesHLA B-27 Associated Anterior Uveitis: American Academy of Optometry: Case Report #1Shabrina Amalia SuciNo ratings yet
- Associations of High Hypermetropia in Childhood JE Marr Et AlDocument2 pagesAssociations of High Hypermetropia in Childhood JE Marr Et AlArisa RosyadaNo ratings yet
- RiddlesDocument44 pagesRiddlessafasayed100% (1)
- Case Report: Vertigo As A Predominant Manifestation of NeurosarcoidosisDocument5 pagesCase Report: Vertigo As A Predominant Manifestation of NeurosarcoidosisDjumadi AkbarNo ratings yet
- Grand Rounds Glaucoma NTGDocument37 pagesGrand Rounds Glaucoma NTGRon TongbaiNo ratings yet
- Acute Multifocal Placoid Pigment EpitheliopathyDocument8 pagesAcute Multifocal Placoid Pigment EpitheliopathyremotmNo ratings yet
- NTG Practical ApproachDocument6 pagesNTG Practical ApproachRasha Mounir Abdel-Kader El-TanamlyNo ratings yet
- Subhyaloid Hemorrhage in Cerebral Malaria: Case ReportDocument4 pagesSubhyaloid Hemorrhage in Cerebral Malaria: Case ReportFaza KahfiNo ratings yet
- Clinical Challenges: Not Again!Document8 pagesClinical Challenges: Not Again!nimmma_3No ratings yet
- Dafpus 10 Sympathetic - Opthalmia-2Document7 pagesDafpus 10 Sympathetic - Opthalmia-2Anonymous jh87ryNo ratings yet
- Orbital Apex Syndrome from Herpes Zoster OphthalmicusDocument4 pagesOrbital Apex Syndrome from Herpes Zoster OphthalmicusYosiita KartinaaNo ratings yet
- Progressive Multifocal Leukoencephalopathy Case ReportDocument5 pagesProgressive Multifocal Leukoencephalopathy Case Reportnoorhadi.n10No ratings yet
- Pi Is 0161642099901604Document3 pagesPi Is 0161642099901604Wahyu SholekhuddinNo ratings yet
- Neurology and Neurotherapy: ClinmedDocument4 pagesNeurology and Neurotherapy: ClinmedChristian Hasudungan NainggolanNo ratings yet
- Pratique: Understanding The Patient With Epilepsy and Seizures in The Dental PracticeDocument6 pagesPratique: Understanding The Patient With Epilepsy and Seizures in The Dental PracticeJade M. LolongNo ratings yet
- Reversible Ocular Toxicity Related To Tamoxifen Therapy: DE) Also of ofDocument3 pagesReversible Ocular Toxicity Related To Tamoxifen Therapy: DE) Also of ofsafasayedNo ratings yet
- Bilateral Vision Loss Associated With Radiofrequency ExposureDocument5 pagesBilateral Vision Loss Associated With Radiofrequency ExposurePisj Belongs To WhatsappNo ratings yet
- Practice Parameter The Role of Corticosteroids in The Management of Acute Monosymptomatic Optic NeuritisDocument9 pagesPractice Parameter The Role of Corticosteroids in The Management of Acute Monosymptomatic Optic Neuritissatyagraha84No ratings yet
- Optic Neuritis: Clinical PR ActiceDocument8 pagesOptic Neuritis: Clinical PR ActiceafbmgNo ratings yet
- Recurrent Optic Neuritis As A Clinical Manifestation in Multiple Sclerosis A Case Report - Anisa Feby ArifaniDocument11 pagesRecurrent Optic Neuritis As A Clinical Manifestation in Multiple Sclerosis A Case Report - Anisa Feby Arifanidenny070111209No ratings yet
- American Journal of Ophthalmology Case Reports: Yael Sharon, David S. Chu TDocument5 pagesAmerican Journal of Ophthalmology Case Reports: Yael Sharon, David S. Chu TDaviel Quin DavNo ratings yet
- Hemorrhagic Unilateral RetinopathyDocument7 pagesHemorrhagic Unilateral RetinopathyHanna_RNo ratings yet
- Glaucoma - State of The Art and Perspectives On TreatmentDocument17 pagesGlaucoma - State of The Art and Perspectives On TreatmentbarbaraNo ratings yet
- Test Utilization and Value in The Evaluation of Peripheral NeuropathiesDocument8 pagesTest Utilization and Value in The Evaluation of Peripheral NeuropathiesFlea CidNo ratings yet
- Neuroretinitis Syphilis in Human Immunodeficiency Virus-Infected PatientDocument6 pagesNeuroretinitis Syphilis in Human Immunodeficiency Virus-Infected PatientAnindya AgrasidiNo ratings yet
- Treatment of Age-Related Macular Degeneration: Risk FactorsDocument4 pagesTreatment of Age-Related Macular Degeneration: Risk FactorsisraelrtNo ratings yet
- Care 2008 38-60Document30 pagesCare 2008 38-60yateema4ever100% (2)
- Uveitis Diagnosis and Treatment GuideDocument72 pagesUveitis Diagnosis and Treatment GuideSafa Abdualrahaman Ali HamadNo ratings yet
- A Painful Red EyeDocument2 pagesA Painful Red EyeCristhian Agustin ParedesNo ratings yet
- Qstream-1 2Document55 pagesQstream-1 2Vijay100% (1)
- Visual Evoked Potentials in Guillain-Barré Syndrome: MethodsDocument6 pagesVisual Evoked Potentials in Guillain-Barré Syndrome: MethodsYulianti PurnamasariNo ratings yet
- Treatment of Optic Neuritis With Erythropoietin (TONE) : A Randomised, Double-Blind, Placebo-Controlled Trial - Study ProtocolDocument10 pagesTreatment of Optic Neuritis With Erythropoietin (TONE) : A Randomised, Double-Blind, Placebo-Controlled Trial - Study ProtocolDyah Farah DetaNo ratings yet
- Bell's Palsy - Treatment GuidelinesDocument4 pagesBell's Palsy - Treatment GuidelinessevattapillaiNo ratings yet
- Case Discussion SLEDocument4 pagesCase Discussion SLERanjeetha SivaNo ratings yet
- Cidp Nejm PDFDocument14 pagesCidp Nejm PDFFebrianaNo ratings yet
- The Pathophysiology and Treatment of Glaucoma A ReviewDocument11 pagesThe Pathophysiology and Treatment of Glaucoma A ReviewJacqueline LozzNo ratings yet
- Traumatic Hyphema: A Teaching Case Report: Priscilla Lenihan, OD Dorothy Hitchmoth, OD, FAAODocument9 pagesTraumatic Hyphema: A Teaching Case Report: Priscilla Lenihan, OD Dorothy Hitchmoth, OD, FAAOMarshaNo ratings yet
- Unilateral Retinitis Pigmentosa Review: Causes, Imaging, DiagnosisDocument5 pagesUnilateral Retinitis Pigmentosa Review: Causes, Imaging, DiagnosisPriya ChandrasekaranNo ratings yet
- No Laughing Matter: Subacute Degeneration of The Spinal Cord Due To Nitrous Oxide InhalationDocument7 pagesNo Laughing Matter: Subacute Degeneration of The Spinal Cord Due To Nitrous Oxide InhalationgshjfkNo ratings yet
- 100255-Article Text-1482-2-10-20220311Document6 pages100255-Article Text-1482-2-10-20220311Fachry RahmanNo ratings yet
- Neurosyphilis Masquerading As An Acute Adie's Tonic Pupil: Report of A CaseDocument6 pagesNeurosyphilis Masquerading As An Acute Adie's Tonic Pupil: Report of A CaseOlvaria MisfaNo ratings yet
- Case Report: Unilateral Optic Neuropathy and Acute Angle-Closure Glaucoma Following Snake EnvenomationDocument5 pagesCase Report: Unilateral Optic Neuropathy and Acute Angle-Closure Glaucoma Following Snake EnvenomationErnesErlyanaSuryawijayaNo ratings yet
- Lecture 1 PDFDocument3 pagesLecture 1 PDFIvana BeatriceNo ratings yet
- Allopurinol-Induced Drug Reactions With Eosinophilia and Systemic Symptoms Syndrome With Interstitial NephritisDocument12 pagesAllopurinol-Induced Drug Reactions With Eosinophilia and Systemic Symptoms Syndrome With Interstitial NephritisJoc HerreraNo ratings yet
- Clinical Diagnosis and Management of Ocular Myasthenia GravisDocument11 pagesClinical Diagnosis and Management of Ocular Myasthenia GravisAinul NurrahmahNo ratings yet
- Chen2014 Jurnal MataDocument3 pagesChen2014 Jurnal MatanidaNo ratings yet
- Driving PressureDocument9 pagesDriving PressureHydr OrtgaNo ratings yet
- 50 Apnea Test For Determination of Clinical Brain Death1Document1 page50 Apnea Test For Determination of Clinical Brain Death1Maximiliano Vielma RojasNo ratings yet
- 04 Enteral FeedingDocument4 pages04 Enteral FeedingRose SedanzaNo ratings yet
- Ascitis Matters PDFDocument11 pagesAscitis Matters PDFckeshavaNo ratings yet
- ProcedureDocument1 pageProcedureckeshavaNo ratings yet
- Ascitis Matters PDFDocument11 pagesAscitis Matters PDFckeshavaNo ratings yet
- Crypto PDFDocument3 pagesCrypto PDFckeshavaNo ratings yet
- Aen017 PDFDocument10 pagesAen017 PDFckeshavaNo ratings yet
- Interventional Spine and Pain Procedures in Patients On Antiplatelet and Anticoagulant Medications (Second Edition)Document38 pagesInterventional Spine and Pain Procedures in Patients On Antiplatelet and Anticoagulant Medications (Second Edition)ckeshavaNo ratings yet
- 30559228: 2. Classification and Diagnosis of Diabetes Standards of Medical Care in Diabetes-2019 PDFDocument16 pages30559228: 2. Classification and Diagnosis of Diabetes Standards of Medical Care in Diabetes-2019 PDFMic AtefNo ratings yet
- Platinum Aura Edge Benefit GuideDocument20 pagesPlatinum Aura Edge Benefit GuideNaveen KumarNo ratings yet
- Head Rules Over The Heart: Cardiac Manifestations of Cerebral DisordersDocument7 pagesHead Rules Over The Heart: Cardiac Manifestations of Cerebral DisordersckeshavaNo ratings yet
- Renal Artery StenosisDocument6 pagesRenal Artery StenosisckeshavaNo ratings yet
- BkaDocument4 pagesBkackeshavaNo ratings yet
- How Rich Neighbours Can Make Us Unhappy: Weekends Keep Changing With TimeDocument1 pageHow Rich Neighbours Can Make Us Unhappy: Weekends Keep Changing With TimeckeshavaNo ratings yet
- Karnataka Dental College Fees Structure 2018Document4 pagesKarnataka Dental College Fees Structure 2018samiNo ratings yet
- I Still Have The Bat Yuvraj Hit Six Sixes With': 7 Smart Accessories To Jazz Up Your TabletDocument1 pageI Still Have The Bat Yuvraj Hit Six Sixes With': 7 Smart Accessories To Jazz Up Your TabletckeshavaNo ratings yet
- Should you buy your dream car nowDocument1 pageShould you buy your dream car nowckeshavaNo ratings yet
- SC Upholds Law Making PAN Linking With Aadhaar Must: Investors: Infy Share Sale No Bad Thing'Document1 pageSC Upholds Law Making PAN Linking With Aadhaar Must: Investors: Infy Share Sale No Bad Thing'ckeshavaNo ratings yet
- I Still Have The Bat Yuvraj Hit Six Sixes With': 7 Smart Accessories To Jazz Up Your TabletDocument1 pageI Still Have The Bat Yuvraj Hit Six Sixes With': 7 Smart Accessories To Jazz Up Your TabletckeshavaNo ratings yet
- Twin Frenzy: George, Amal Welcome Twins... Beyonce You'Re NextDocument1 pageTwin Frenzy: George, Amal Welcome Twins... Beyonce You'Re NextckeshavaNo ratings yet
- A Taste of Exotic, Imported Fruits: Summer Is Here, and Bengaluru GetsDocument1 pageA Taste of Exotic, Imported Fruits: Summer Is Here, and Bengaluru GetsckeshavaNo ratings yet
- Cafes Cut Wi-Fi Connection To Get You Chatting: Statin Side-Effects Imagined?Document1 pageCafes Cut Wi-Fi Connection To Get You Chatting: Statin Side-Effects Imagined?ckeshavaNo ratings yet
- FB Tech Can Make Webcam Spy On You: Nasa's New Rover Is A Cool Batmobile' Intimate Grooming For Men On The RiseDocument1 pageFB Tech Can Make Webcam Spy On You: Nasa's New Rover Is A Cool Batmobile' Intimate Grooming For Men On The RiseckeshavaNo ratings yet
- A Taste of Exotic, Imported Fruits: Summer Is Here, and Bengaluru GetsDocument1 pageA Taste of Exotic, Imported Fruits: Summer Is Here, and Bengaluru GetsckeshavaNo ratings yet
- 52 near misses in Indian skies since 2014 as minister's son dies in Mercedes crashDocument1 page52 near misses in Indian skies since 2014 as minister's son dies in Mercedes crashckeshavaNo ratings yet
- Kinds of Travellers We Meet in Our LifetimeDocument1 pageKinds of Travellers We Meet in Our LifetimeckeshavaNo ratings yet
- SC Upholds Law Making PAN Linking With Aadhaar Must: Investors: Infy Share Sale No Bad Thing'Document1 pageSC Upholds Law Making PAN Linking With Aadhaar Must: Investors: Infy Share Sale No Bad Thing'ckeshavaNo ratings yet
- CPG Management DiabetesDocument49 pagesCPG Management DiabetesEmi Tri SiswantiNo ratings yet
- Mononeuropathy MultiplexDocument2 pagesMononeuropathy MultiplexElena EllaNo ratings yet
- Guillain-Barré SyndromeDocument60 pagesGuillain-Barré SyndromekharaNo ratings yet
- Increase Air Element with Gyan MudraDocument9 pagesIncrease Air Element with Gyan MudraKr SelvakumarNo ratings yet
- Review: New Horizons in Diabetic Neuropathy: Mechanisms, Bioenergetics, and PainDocument18 pagesReview: New Horizons in Diabetic Neuropathy: Mechanisms, Bioenergetics, and PainDIBA TRIULANDARI KUSNADINo ratings yet
- Nri2022 8336561Document14 pagesNri2022 8336561Carolina SidabutarNo ratings yet
- Disorders of The Nervous System: Vascular DisorderDocument3 pagesDisorders of The Nervous System: Vascular DisorderHetty100% (1)
- Axinerve NP MonographDocument12 pagesAxinerve NP MonographShama Safiana100% (1)
- Clinical Approach To QuadriplegiaDocument11 pagesClinical Approach To QuadriplegiaUmesh BabuNo ratings yet
- Slide Chronic Pain-Rudy Hidayat-Iai PDFDocument30 pagesSlide Chronic Pain-Rudy Hidayat-Iai PDFAsri ArrachmanNo ratings yet
- Diabetes MellitusDocument8 pagesDiabetes MellitusJaja RamosNo ratings yet
- Understanding Neuropati Perifer and Its CausesDocument28 pagesUnderstanding Neuropati Perifer and Its CausesDjumadi AkbarNo ratings yet
- Diabetes Complication LectureDocument63 pagesDiabetes Complication LectureDoni MarthenNo ratings yet
- Acupuncture and Cancer Pain: Yan Cui Magram and Gary E. DengDocument4 pagesAcupuncture and Cancer Pain: Yan Cui Magram and Gary E. DengirmaNo ratings yet
- EUPO 2016: Course On Neuro-Ophthalmology and StrabismusDocument180 pagesEUPO 2016: Course On Neuro-Ophthalmology and StrabismusJulio EHNo ratings yet
- Faraz Pearls MRCP Part 2 by Faraz Ahmed YnzDocument466 pagesFaraz Pearls MRCP Part 2 by Faraz Ahmed Ynzashwini dhote100% (1)
- Emg Conceptos - Nerve Conduction StudiesDocument23 pagesEmg Conceptos - Nerve Conduction StudiesDeborah SalinasNo ratings yet
- CV for Neurologist Specializing in PainDocument26 pagesCV for Neurologist Specializing in Painmelmartana100% (2)
- Palliative Care in Multiple MyelomaDocument45 pagesPalliative Care in Multiple MyelomamadvincyNo ratings yet
- 5502 PSA Annual Gold Questionnaire 2018 v3Document8 pages5502 PSA Annual Gold Questionnaire 2018 v3Abdul B. Gharaibeh100% (1)
- Best Neurologist in BangaloreDocument3 pagesBest Neurologist in BangaloreAlgoTrends AlgoTrendsNo ratings yet
- NPMDS assessmentDocument8 pagesNPMDS assessmentF4ARNo ratings yet
- Brachial NeuritisDocument7 pagesBrachial Neuritisfirza yoga baskoroNo ratings yet
- Managing Diabetic NeuropathyDocument38 pagesManaging Diabetic NeuropathyAnanda Christie AngelinNo ratings yet
- 200122117aravind PDFDocument109 pages200122117aravind PDFvikas mishraNo ratings yet
- Callaghan 2015Document10 pagesCallaghan 2015ardyariniNo ratings yet