Professional Documents
Culture Documents
Chemical Basis of Life
Uploaded by
Chris_Barber09Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Chemical Basis of Life
Uploaded by
Chris_Barber09Copyright:
Available Formats
CLEP Biology - Core Concept Cheat Sheet
02: Chemical Basis of Life
Key Terms
Organic Chemicals
Atoms:
o Electrons: both energy and substance particles
o Neutrons
o Protons
Classified by the functional groups:
Alcohols, R-OH
Aldehydes, R-CHO, R-C=O
|
O
H
||
Ketones, R-C-R, R-CO-R
O
||
Carboxylic Acids, R-COOH, R-C-OH
Amines, R-NH2, R-N-H
|
H
Molecules:
o Formed by atoms
o Joined by chemical bonds
o Molecular formula and structure formula
Organic Molecules/macromolecules:
o Amino acids --> proteins
o Monosacchrides --> polysaccharides
o Fatty acids --> lipids
o Nucleotides --> nucleic acids
Isotope: Atoms have same proton numbers but may differ
in neutron numbers
Energy shell: Electrons occupy orbital around nucleus, these
are called energy shells. The most inner shell (K) contains 2
electron maximum, the L and M shell contain 8 maximum
each.
Organic chemicals: Chemicals are made from living
organisms and contain carbon backbones.
Isomers: Chemicals that have same molecular formula but
different structure formula.
Buffers: Solutions which resist change in pH upon addition
of small amounts of acid or base.
Electrolytes: Chemicals that can release ions into solutions
pH: pH represents the concentration of hydrogen ions [H+] in
solution.
pH = -log [H+]
Enzymes: Proteins that serve as catalysts for biochemical
reactions.
Enthropy: A measure for a system's degree of disorder. It
increases with increasing disorder.
Law of thermodynamics:
The first Law: The total energy of the universe is always
conserved. Energy can neither be created nor destroyed.
The second Law: The universe tends towards maximum
disorder, or, in other words: the direction of all
spontaneous processes is such as to increase the
entropy of a system plus its surroundings
G: Change of free energy of a system.
G negative reaction: spontaneous
G positive reaction: non-spontaneous
Chemical Bonds
O|
Organic Phosphates, R-OPO32-, R-O-P=O
|
OThiols, R-SH
Important Biochemical Molecules
Organic Molecules/macromolecules:
o
o
Polysaccharides
Monomer unit: monosaccharide
Store energy, provide building unit
o
o
Lipids
Monomer unit: fatty acids, glycerol
Store energy, membrane construction, hormones
o
o
Proteins:
Monomer unit: amino acids
Structure protein, enzymes
o
o
Nucleic Acids:
Monomer unit: nucleotides
Genetic material
Chemical Reactions
Coupled reactions: Many biosynthesis reactions are coupled
to ATP hydrolysis which can provide energy and therefore the
overall reaction can be delta G negative.
G negative reaction: spontaneous
G positive reaction: non-spontaneous
Enzyme catalyzed reactions: Lower the activation free
energy but do not change the G.
Biochemical Reaction Types and Enzymes
Oxidation-reduction reactions: oxidoreductase
Chemical bonds store energy. For covalent bonds, the more
electrons a bond share, the more energy it stores.
Ionic bond: ionic bond forms when atoms lose or gain
electrons.
Covalent bond: Covalent bonds form when atoms share
electrons, very strong bonds. The major one in organic
chemicals.
Hydrogen bond: Weak electrical attraction between the
positive end of one molecule and the negative end of another
Intramolecular or intermolecular functional group-transfer
reactions: transfease
Hydrolysis of esters, ethers, and amides: hydrolase
Elimination or addition reactions: lyase.
Isomerization reactions: isomerase
Formation of ester, thiol ester, and amide linkages: ligase
How to Use This Cheat Sheet: These are the keys related this topic. Try to read through it carefully twice then rewrite it out on
a blank sheet of paper. Review it again before the exams.
RapidLearningCenter.com.com
Rapid Learning Inc. All Rights Reserved
You might also like
- Physics Rules 5Document10 pagesPhysics Rules 5Chris_Barber09100% (6)
- Cells Cheat SheetDocument2 pagesCells Cheat SheetUlka Sutar100% (2)
- Surviving Chemistry: A Guided Study Book For High School ChemistryDocument83 pagesSurviving Chemistry: A Guided Study Book For High School ChemistryE3 Scholastic Publishing100% (2)
- Microbiology MasterDocument13 pagesMicrobiology MasterDelixae Phoinix100% (6)
- Mitosis and MeiosisDocument1 pageMitosis and MeiosisChris_Barber09100% (1)
- ImmunologyDocument1 pageImmunologyChris_Barber0986% (7)
- Genetics NotesDocument6 pagesGenetics NotesShane Tang100% (2)
- Chemistry MnemonicsDocument16 pagesChemistry MnemonicsrishikeshkallaNo ratings yet
- VirusesDocument1 pageVirusesChris_Barber09100% (2)
- Biochemistry Notes PDFDocument11 pagesBiochemistry Notes PDFChris_Barber0950% (6)
- 01: Introduction To Evolutionary Thought: Key Evolution TermsDocument1 page01: Introduction To Evolutionary Thought: Key Evolution TermsBlaine Rogalski100% (2)
- ProteinsDocument1 pageProteinsChris_Barber09No ratings yet
- Jack Westin MCAT Content BiochemistryDocument52 pagesJack Westin MCAT Content BiochemistryLora100% (2)
- Cell Biology IntroductionDocument1 pageCell Biology IntroductionChris_Barber09100% (1)
- RP Diet Template FAQ 2Document23 pagesRP Diet Template FAQ 2Schuyler Brass100% (1)
- Organic Chem Midterm 1 Multi+keyDocument1 pageOrganic Chem Midterm 1 Multi+keyNorma Leticia RamosNo ratings yet
- Cheat Sheet For Organic Chemistry Midterm 1 2015 - 1Document1 pageCheat Sheet For Organic Chemistry Midterm 1 2015 - 1Norma Leticia Ramos33% (3)
- Molecular Cell Biology MasterDocument6 pagesMolecular Cell Biology Masterdealt100% (5)
- MCAT Organic Summary SheetDocument6 pagesMCAT Organic Summary SheetSpencer Thomas100% (2)
- Naming Organic MoleculesDocument47 pagesNaming Organic MoleculesSandeep BadarlaNo ratings yet
- Amino Acid NotesDocument15 pagesAmino Acid NotesChris_Barber09No ratings yet
- Chemistry Cheat SheetDocument10 pagesChemistry Cheat Sheetbrook92% (39)
- Bio Cheat Sheet MasterDocument7 pagesBio Cheat Sheet MasterChris_Barber0986% (7)
- Chemistry MasterDocument7 pagesChemistry MasterDelixae Phoinix100% (2)
- Anatomy and PhysiologyDocument6 pagesAnatomy and PhysiologyDelixae Phoinix80% (5)
- Amino Acids & Proteins PDFDocument135 pagesAmino Acids & Proteins PDFJoelle AwarNo ratings yet
- Protein Synthesis Notes PDFDocument3 pagesProtein Synthesis Notes PDFChris_Barber09No ratings yet
- MCAT Biology Notes 2 PDFDocument23 pagesMCAT Biology Notes 2 PDFChris_Barber09100% (1)
- CHEM Cheat Sheet 3Document2 pagesCHEM Cheat Sheet 3Ruby Rodriguez100% (2)
- Practical Biology: For Advanced Level, Medical and Intermediate StudentsFrom EverandPractical Biology: For Advanced Level, Medical and Intermediate StudentsNo ratings yet
- Nmat Biology Cell Biology 1.1 Eukaryotic & Prokaryotic CellsDocument12 pagesNmat Biology Cell Biology 1.1 Eukaryotic & Prokaryotic Cellssavina100% (1)
- Physics2A CheatSheetDocument1 pagePhysics2A CheatSheetNastassja LopezNo ratings yet
- BiochemistryDocument383 pagesBiochemistrybalajimeie92% (12)
- MitochondriaDocument6 pagesMitochondriakevin smithNo ratings yet
- Autonomic Nervous SystemDocument1 pageAutonomic Nervous SystemChris_Barber09No ratings yet
- Atomic and Molecular Physics (PDFDrive)Document216 pagesAtomic and Molecular Physics (PDFDrive)Vidvata - Learning boosters as technologyNo ratings yet
- MCAT MetabolismDocument4 pagesMCAT MetabolismNawledge9308100% (1)
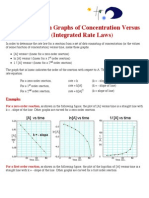
- Rate Law GraphsDocument2 pagesRate Law GraphsChris_Barber09No ratings yet
- Rate Law GraphsDocument2 pagesRate Law GraphsChris_Barber09No ratings yet
- MCAT Biology Notes 3 PDFDocument16 pagesMCAT Biology Notes 3 PDFChris_Barber09No ratings yet
- Phylogenetic Trees Click Learn WorksheetDocument4 pagesPhylogenetic Trees Click Learn Worksheetfcm31450% (1)
- College PhysicsDocument6 pagesCollege PhysicsDelixae Phoinix100% (1)
- 02 Protein DigestionDocument44 pages02 Protein DigestionJohn MusaNo ratings yet
- OCR Biology F211 Cells, Exchange and Transport Revision NotesDocument4 pagesOCR Biology F211 Cells, Exchange and Transport Revision Notes24wrightphilip100% (1)
- BIOCHEMISTRY, CELL AND MOLECULAR BIOLOGY: Passbooks Study GuideFrom EverandBIOCHEMISTRY, CELL AND MOLECULAR BIOLOGY: Passbooks Study GuideNo ratings yet
- Human Physiology IntroductionDocument1 pageHuman Physiology IntroductionChris_Barber09No ratings yet
- BIOL 200 Molecular Biology Lecture NotesDocument44 pagesBIOL 200 Molecular Biology Lecture NotesDantong JiaNo ratings yet
- Organic Chemistry NotesDocument9 pagesOrganic Chemistry NotesBuana SandilaNo ratings yet
- Notes CarbohydratesDocument21 pagesNotes CarbohydratesChris_Barber09100% (1)
- Notes CarbohydratesDocument21 pagesNotes CarbohydratesChris_Barber09100% (1)
- Microbiology NotesDocument177 pagesMicrobiology NotesMarichu Rogas91% (11)
- Idli AssignmentDocument20 pagesIdli AssignmentJasz Azliey Azzam100% (1)
- AP Bio EnzymesDocument50 pagesAP Bio Enzymesjulie raines100% (6)
- Muscular SystemDocument1 pageMuscular SystemChris_Barber09100% (1)
- Peripheral Nervous SystemDocument1 pagePeripheral Nervous SystemChris_Barber09No ratings yet
- Urinary SystemDocument1 pageUrinary SystemChris_Barber09100% (1)
- Biotechnoloy Introduction and Application: Table of ContentDocument40 pagesBiotechnoloy Introduction and Application: Table of ContentnescafeforeverNo ratings yet
- Bacteria: Type of Food Depends On Organism)Document5 pagesBacteria: Type of Food Depends On Organism)Chris_Barber09No ratings yet
- Biology NotesDocument6 pagesBiology NotesElizaNo ratings yet
- Genetic Cheat SheetDocument11 pagesGenetic Cheat SheetLeah MaloneyNo ratings yet
- Preliminary Instructions: Cu Ni CR Fe FeDocument4 pagesPreliminary Instructions: Cu Ni CR Fe FeEmmanuel Ryan100% (1)
- 16: Work, Power, and Energy: Key Physics Terms Key ConceptsDocument1 page16: Work, Power, and Energy: Key Physics Terms Key ConceptsBlaine RogalskiNo ratings yet
- Introduction To Biology ReviewerDocument22 pagesIntroduction To Biology ReviewerAnton Miguel Jordan83% (6)
- Phylogenetic Tree Creation Morphological and Molecular Methods For 07-JohnsonDocument35 pagesPhylogenetic Tree Creation Morphological and Molecular Methods For 07-JohnsonCHRISTEROP100% (2)
- AP Biology - Chapter 30 Discussion AnswersDocument3 pagesAP Biology - Chapter 30 Discussion Answersangel91me6371100% (6)
- Zoology Notes Class 11Document109 pagesZoology Notes Class 11Suresh chand50% (2)
- Molecular Biology of The Cell Cumulative Final Exam Study GuideDocument22 pagesMolecular Biology of The Cell Cumulative Final Exam Study Guiderazgriz1211No ratings yet
- Audio Osmosis - Organic ChemistryDocument9 pagesAudio Osmosis - Organic ChemistryddNo ratings yet
- Lecture Notes On Basic Chemistry and Biochemistry (Chapter 2)Document10 pagesLecture Notes On Basic Chemistry and Biochemistry (Chapter 2)Helen ShuangNo ratings yet
- Chapter 2 Chemical Bsis of LifeDocument9 pagesChapter 2 Chemical Bsis of LifeMary Ann SacramentoNo ratings yet
- Biochemistry Fundamentals: An Overview of Enzymes, Energy and BioenergeticsDocument58 pagesBiochemistry Fundamentals: An Overview of Enzymes, Energy and BioenergeticsMarc Imhotep Cray, M.D.100% (1)
- Chapter 2 - Levels of Organization of The Human BodyDocument9 pagesChapter 2 - Levels of Organization of The Human BodyFrancine Danielle TiponesNo ratings yet
- Macromolecules PDFDocument11 pagesMacromolecules PDFChris_Barber09No ratings yet
- Nucleic Acids and Their Structure: Information?Document4 pagesNucleic Acids and Their Structure: Information?Chris_Barber09No ratings yet
- Reproductive System NotesDocument3 pagesReproductive System NotesChris_Barber09No ratings yet
- Heart Notes PDFDocument2 pagesHeart Notes PDFChris_Barber09No ratings yet
- Bacteria: Type of Food Depends On Organism)Document5 pagesBacteria: Type of Food Depends On Organism)Chris_Barber09No ratings yet
- Cardiovasculary System PDFDocument5 pagesCardiovasculary System PDFChris_Barber09No ratings yet
- Digestive System PDFDocument3 pagesDigestive System PDFChris_Barber09No ratings yet
- MCAT O-Chem NotesDocument1 pageMCAT O-Chem NotesChris_Barber09No ratings yet
- Extra Chirality ProblemsDocument21 pagesExtra Chirality ProblemsChris_Barber09No ratings yet
- Aromatic Notes 2 PDFDocument6 pagesAromatic Notes 2 PDFChris_Barber09100% (1)
- CARBSDocument24 pagesCARBSGulus CfNo ratings yet
- Chapter 16:substituent Effects in Aromatic SubstitutionDocument2 pagesChapter 16:substituent Effects in Aromatic SubstitutionChris_Barber09No ratings yet
- Physics Rules 2Document4 pagesPhysics Rules 2Chris_Barber09No ratings yet
- MCAT Physics Equation SheetDocument6 pagesMCAT Physics Equation SheetChris_Barber09No ratings yet
- Regents Physics Exam Prep: 101 Facts You Should Know: MechanicsDocument3 pagesRegents Physics Exam Prep: 101 Facts You Should Know: MechanicsChris_Barber09No ratings yet
- AAMC 9 Essay 1Document4 pagesAAMC 9 Essay 1Chris_Barber09No ratings yet
- Sample Essay #1: Education Makes Everyone EqualDocument4 pagesSample Essay #1: Education Makes Everyone EqualChris_Barber09No ratings yet
- 107 Rules in PhysicsDocument2 pages107 Rules in PhysicsChris_Barber09No ratings yet
- Physics Rules 1Document2 pagesPhysics Rules 1Chris_Barber09No ratings yet
- Advanced Placement Physics Physics OverviewDocument9 pagesAdvanced Placement Physics Physics OverviewChris_Barber09No ratings yet
- Sample Essay #1: Advancements in Communication Technology Have Reduced The Quality of Human InteractionDocument6 pagesSample Essay #1: Advancements in Communication Technology Have Reduced The Quality of Human InteractionChris_Barber09No ratings yet
- Essay #1: A Politician's Lifestyle Should Reflect His or Her Political ViewsDocument4 pagesEssay #1: A Politician's Lifestyle Should Reflect His or Her Political ViewsChris_Barber09No ratings yet
- Peripheral Nervous SystemDocument1 pagePeripheral Nervous SystemChris_Barber09No ratings yet
- Sample Essay #1: Progress Often Complicates As Much As It SimplifiesDocument4 pagesSample Essay #1: Progress Often Complicates As Much As It SimplifiesChris_Barber09No ratings yet
- Animal GluesDocument12 pagesAnimal GluesLucas SilveiraNo ratings yet
- Channa Striata PDFDocument9 pagesChanna Striata PDFM Husni MubarokNo ratings yet
- Question Bank IX CH-5 CELL - THE FUNDAMENTAL UNIT OF LIFEDocument18 pagesQuestion Bank IX CH-5 CELL - THE FUNDAMENTAL UNIT OF LIFENoel MammenNo ratings yet
- A Novel Technique To Evaluate Nail Softening Effects of Different Urea FormulationsDocument10 pagesA Novel Technique To Evaluate Nail Softening Effects of Different Urea FormulationsHiep X NguyenNo ratings yet
- Shuguang Zhang Et Al - Design of Nanostructured Biological Materials Through Self-Assembly of Peptides and ProteinsDocument8 pagesShuguang Zhang Et Al - Design of Nanostructured Biological Materials Through Self-Assembly of Peptides and ProteinsGmewop30m100% (1)
- Unit IiDocument51 pagesUnit IiNageswari SowmiNo ratings yet
- Polymers 15 00608 v3Document23 pagesPolymers 15 00608 v3FernandaNo ratings yet
- Chapter+5 19+McKee+Suggested+ProblemsDocument30 pagesChapter+5 19+McKee+Suggested+ProblemsShivam PatelNo ratings yet
- Laboratory Exercise No. 4 Qualitative Protein AnalysesDocument5 pagesLaboratory Exercise No. 4 Qualitative Protein AnalysesErikaJoillePatayonNo ratings yet
- Protien ASDocument25 pagesProtien ASRabia RafiqueNo ratings yet
- Cell Organelles and Its Primary FunctionsDocument3 pagesCell Organelles and Its Primary FunctionsSuzie Ann100% (1)
- Where Did Life Come From (An Essay)Document3 pagesWhere Did Life Come From (An Essay)Yuan Iñigo PulidoNo ratings yet
- AP BioDocument4 pagesAP BioAlisha MasonNo ratings yet
- Production of Pharmaceutical Compounds Through Microbial FermentationDocument2 pagesProduction of Pharmaceutical Compounds Through Microbial FermentationSAFC-Global100% (1)
- Macromolecule LabDocument7 pagesMacromolecule Labapi-318665838No ratings yet
- Biotechnology Eligibility Test (BET) For DBT-JRF Award (2011-12)Document20 pagesBiotechnology Eligibility Test (BET) For DBT-JRF Award (2011-12)Nandakumar HaorongbamNo ratings yet
- Chapter Test A: BiochemistryDocument5 pagesChapter Test A: Biochemistrykatelyn mcdonoughNo ratings yet
- Protein Terapetik, Vaksin Dan Teknologi Produksi Protein RekombinanDocument23 pagesProtein Terapetik, Vaksin Dan Teknologi Produksi Protein Rekombinan19 095 Gina Nafsiah PutriNo ratings yet
- 1 - Lecture - A Proteostasis Concept - B - Protein Structure C - Bioanalytics X Ray Crystalography - 1-20 - HandoutDocument22 pages1 - Lecture - A Proteostasis Concept - B - Protein Structure C - Bioanalytics X Ray Crystalography - 1-20 - HandoutHamud ShmalyNo ratings yet
- Tura KuDocument104 pagesTura KuFitzgerald BillyNo ratings yet
- (Doi 10.1016 - J.physa.2016.08.033) PDFDocument27 pages(Doi 10.1016 - J.physa.2016.08.033) PDFGizwa AiichaicchapratiwieNo ratings yet