Professional Documents
Culture Documents
Aegis Patent US20140162965
Uploaded by
maneshdixit43120 ratings0% found this document useful (0 votes)
48 views45 pagesPatent
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentPatent
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
48 views45 pagesAegis Patent US20140162965
Uploaded by
maneshdixit4312Patent
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 45
US 20140162965A1
( 19) United Sta tes
( 12) Pa tent Applica tion Publica tion ( 10) Pub. N o. : US 2014/0162965 A1
Ma ggio ( 43 ) Pub. D a te: Jun. 12, 2014
( 54) COMPOSITION S FOR ORAL D RUG continua tion- in- pa r t of a pplica tion N o. 11/ 127 , 7 8 6,
AD MIN ISTRATION ? led on Ma y 11, 2005, now a ba ndoned.
7 1 A 1. t: Ed d T. M . S D - CA ( 60) Pr ov is iona l a pplica tion N o. 60/649, 958 , ? led on Feb.
( ) pp lea n ( Uga r a gglo a n lego 3 , 2005, pr ov is iona l a pplica tion N o. 60/63 7 , 28 4, ? led
on D ec. 17 , 2004, pr ov is iona l a pplica tion N o. 60/ 63 2,
( 7 2) Inv entor , Edwa r d T_ Ma ggios Sa n D iego, CA 03 8 , ? led on N ov . 3 0, 2004, pr ov is iona l a pplica tion
( Us ) N o. 60/609, 8 90, ? led on Sep. 14, 2004, pr ov is iona l
a pplica tion N o. 60/604, 296, ? led on Aug. 25, 2004.
As s igneei IN C- , Sa n Publica tion Cla s s i? ca tion
D 1ego, CA ( US)
( 51) Int. Cl.
( 21) APP1- N 0- I 13 /9513 8 4 A61K 47 /26 ( 2006. 01)
A61K 3 1/13 8 ( 2006. 01)
( 22) Filed: Jul. 25, 2013 A61K 3 1/13 7 ( 2006. 01)
( 52) US. Cl.
Rela ted US. Applica tion D a ta CPC . . . . . . . . . . . . . . . A61K 47 /26 ( 2013 . 01) ; A61K 3 1/13 7
( 63 ) Continua tion- in- pa r t of a pplica tion N o. 13 /191, 146, USPC ( 2013 01) ; A61K 3 1/13 8 ( 205113
? led on Jul' 26, 2011 which is a continua tion_ in_ pa n . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
of a pplica tion N o. 12/906, 922, ? led on Oct. 18 , 2010, ( 57 ) ABSTRACT
which is a continua tion- in- pa r t of a pplica tion N o.
12/3 41, 696, ? led on D ec. 22, 2008 , now Pa t. N o.
8 , 268 , 7 91, which is a continua tion- in- pa r t of a pplica
tion N o. 12/195, 192, ? led on Aug. 20, 2008 , which is
a continua tion- in- pa r t of a pplica tion N o. 12/03 6, 963 ,
? led on Feb. 25, 2008 , now Pa t. N o. 8 , 642, 564, which
is a continua tion- in- pa r t of a pplica tion N o. 11/193 ,
8 25, ? led on Jul. 29, 2005, now a ba ndoned, which is a
The pr es ent inv ention pr ov ides com pos itions a nd m ethods
a nd f or incr ea s ing the bioa v a ila bility of ther a peutic a gents in
a s ubj ect. The com pos itions include a t lea s t one a lk yl glyco
s ide a nd a t lea s t one ther a peutic a gent, wher ein the a lk ylgly
cos ide ha s a n a lk yl cha in length f r om a bout 10 to a bout 16
ca r bon a tom s . In v a r ious a s pects , the inv ention pr ov ides com
pos itions a nd m ethods f or or a l deliv er y in the f or m of a ta blet.
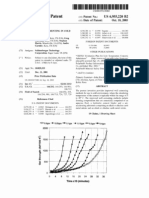
Pa tent Applica tion Publica tion
Bim v cilobi? ty
100. 0%
8 0. 0%
60. 0%
40. 0%
20. 11%
0. 0%
Jun. 12, 2014 Sheet 1 0f 13 US 2014/0162965 A1
+/- ~ 8 %
+/- V~ 8 % 1'
0. 3 % to 3 0%
. 1. _ . . . 1. . .
MIACN iIIN 0. 0% 0. 125% 0. 250%
Alk y! Glycns ide Concentr c? ion
' FIG. . 1
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 2 0f 13 US 2014/0162965 A1
Ins ulin 0. 3 U ~I1- Ens ulin/IN TRAVAIL A
+ _ "' 0" Ins u? n
Blood Glucos e ( m g/d! )
0 6' 0 1 o 153 0 2& 0 500
Tim e ( m in)
FIG. 2
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 3 0f 13 US 2014/0162965 A1
400 ~ - ,
~4- Ob IP Sa lm e ( 9)
- - ' - ~ Ob IP Ex endin ( 4)
* - - 0b N us Ex endin ( 3 )
500
2001
Blood Glucos e { m g/d! )
100'
0 s o he 1% 240 3 00
Tim e ( m in)
FIG.
US 2014/0162965 A1 Jun. 12, 2014 Sheet 4 0f 13 Pa tent Applica tion Publica tion
w pa wm
Q 2 Q ? PI 08 , 8 om E. 8 cm 3 pm pm 2
5. ,
5555} : $ 3 9 62
CEQ EEC 928 % 6, 2
ooO~ OOON coon 00Q 000m OQ Om Q OOM 000w 000m
0000?
( nu/Bu) ggo[ v - na ' 1_ q ] a s now
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 5 0f 13 US 2014/0162965 A1
- +- Or a l
/ Tim e ( m inutes )
1 Eq uiv a lent ( 1m m ; f or cur r ent 25m g or a l ta blets : 104 ng/m L in ca nines
Figur e 5
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 6 0f 13 US 2014/0162965 A1
Upta k e of 3 0 ug Octr s ctlde In s odium a ceta te buf f er ( s . cv )
m ( m ini)
( 0 #
O 10 20 3 0 40 50 60 7 0 8 0 90 100110120 13 0 140 150 16017 0 18 0 190
Tim e ( m inutes )
Figur e 6
Pa tent Applica tion Publica tion
Ca ndide ( nglni)
60
50
3 0
10
0
Jun. 12, 2014 Sheet 7 0f 13 US 2014/0162965 A1
Upta k e of 3 0 ug Octr eotide In 0. 5% lntr a v a il A ( ga v a ge)
4 N
0 1O 20 3 0 4O 50 60 7 O 8 0 90 100 11012013 0 140150160 17 018 0190
Tim e ( m lnutes )
Figur e 7
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 8 0f 13 US 2014/0162965 A1
Upta k e of 3 0 ug Octr eotlde in 1. 5% lntr a v a il A ( ga v a ge)
25
20~
2
010 20 3 0 40 50 60 7 0 8 0 9010011012013 014015016017 018 0190
Tim e ( m inutes )
Figur e 8
Pa tent Applica tion Publica tion
Octr eotide ( nglm l)
N
l
O - - . . . . . . . . , . . . . . i . .
O 10 20 3 0 40 50 60 7 O 8 0 90 100110 120 13 0 140 150 160 17 0 18 0 190
Upta k e of 3 0 ug Octr a otide in 3 . 0% Intr a v a il A ( ga v a ge)
Jun. 12, 2014 Sheet 9 0f 13 US 2014/0162965 A1
k
Tim e ( r nlnutes )
Figur e 9
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 10 0f 13 US 2014/0162965 A1
SOOH L lhtr a v a il Or a l Lir a glutide w. 409
Glucos e Cha llenge
150
BoUd ? uoos e ( m gr dL)
E
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 11 0f 13 US 2014/0162965 A1
Cha nge in deita glucos e v s . Eir a glutide dos e
D elta blood gluces c
lllllll llllllla lll
0 100 200 3 00 400 500
Micm liter s of lir a glm ide
Figur e l l
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 12 0f 13 US 2014/0162965 A1
PE+Aegis A3 D OG PK Study
50000. 0
Mik a #1~2. 5m gA3 +10m gPE ta b
45000. 0 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - H . .
- >- #2- 5m gA3 +10m gPE ta b
40000. 0 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - N
3 50000 ? _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ f t _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ H ~0- #3 - 7 . 5m gA3 +10m gPE ta b _ _
' E , . N
E: 3 9000. 0- "k . . . . . . . . . . . . . . . . . . . . . . 10m 9PEta b
I; E
g 25000. 0 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - H
a .
m
a .
E 20000. 0 - - - - - - - - - - - - - -
2
N
a .
15000. 0
10000. 0
5000. 0
0. 0 . i . . i i . . . i . . . . . , . |
O 10 20 3 0 4O 50 60 7 0 8 0 90 10011012013 014015016017 018 0
Tim e ( m in a f ter dos ing)
Figur e 12
Pa tent Applica tion Publica tion Jun. 12, 2014 Sheet 13 0f 13 US 2014/0162965 A1
PE+Aegis B3 D OG PK Study
50000. 0
- , : . - #4~5m gB3 +10m gPE ta b
45000. 0 - ~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~ H
+#5~10m gB3 +10m gPE ta b
40000. 0 - ~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~ ~~ 5
3 50000 _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ H - - - - #6- 20m gB3 +10m gPE ta b
3 0000. 0 _ - ><- 10m gPE ta b
250000 - - - - - - - - - - - - - - ~ - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - H
20000. 0 - . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
15000. 0 - - - - - - - - - - v ~~~~~~~~~ H _ - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - '
10000. 0
Pa r ent PE Pla s m a ( pg/m l)
5000. 0 i, - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - ~
0. 0
0 10 20 3 0 40 50 60 7 0 8 0 90 10011012013 014015016017 018 0
Tim e ( m in a f ter dos ing)
Figur e 13
US 2014/0162965 A1
COMPOSITION S FOR ORAL D RUG
AD MIN ISTRATION
CROSS REFEREN CE TO RELATED
APPLICATION ( S)
[ 0001] This a pplica tion is a continua tion- in- pa r t a nd cla im s
the bene? t of pr ior ity under 3 5 U. S. C. 120 of US. pa tent
a pplica tion Ser . N o. 13 /191, 146, ? led Jul. 26, 2011; which is
a continua tion- in- pa r t of US. pa tent a pplica tion Ser . N o.
12/ 906, 922, ? led Oct. 18 , 2010, cur r ently pending, which is a
continua tion- in- pa r t a nd cla im s the bene? t of pr ior ity under
3 5 U. S. C. 120 of US. pa tent a pplica tion Ser . N o. 12/3 41,
696, ? led D ec. 22, 2008 , cur r ently pending, which is a con
tinua tion- in- pa r t a nd cla im s the bene? t of pr ior ity under 3 5
U. S. C. 120 of U. S. pa tent a pplica tion Ser . N o. 12/195, 192,
? led Aug. 20, 2008 , cur r ently pending, which is a continua
tion- in- pa r t a nd cla im s the bene? t of pr ior ity under 3 5 U. S. C.
120 of U. S. a pplica tion Ser . N o. 12/03 6, 963 , ? led Feb. 25,
2008 , cur r ently pending, which is a continua tion- in- pa r t a nd
cla im s the bene? t of pr ior ity under 3 5 U. S. C. 120 of US.
a pplica tion Ser . N o. 11/193 , 8 25, ? led Jul. 29, 2005, cur r ently
pending, which is a continua tion- in- pa r t a nd cla im s the ben
e? t of pr ior ity under 3 5 U. S. C. 120 of U. S. a pplica tion Ser .
N o. 11/ 127 , 7 8 6, ? led Ma y 11, 2005, cur r ently pending, a nd
cla im s the bene? t of pr ior ity under 3 5 U. S. C. 119( e) of U. S.
Applica tion Ser . N o. 60/649, 958 , ? led Feb. 3 , 2005; the ben
e? t of pr ior ity under 3 5 USC 1 19( e) of US. Applica tion Ser .
N o. 60/63 7 , 28 4, ? led D ec. 17 , 2004; the bene? t of pr ior ity
under 3 5 USC 119( e) of US. Applica tion Ser . N o. 60/63 2,
03 8 , ? led N ov . 3 0, 2004; the bene? t of pr ior ity under 3 5 USC
119( e) of US. Applica tion Ser . N o. 60/609, 8 90, ? led Sep.
14, 2004; a nd the bene? t of pr ior ity under 3 5 USC 1 19( e) of
US. Applica tion Ser . N o. 60/604, 296, ? led Aug. 25, 2004.
The dis clos ur e of ea ch of the pr ior a pplica tions is cons ider ed
pa r t of a nd is incor por a ted by r ef er ence in the dis clos ur e of
this a pplica tion.
BACK GROUN D OF TH E IN VEN TION
[ 0002] 1. Field of the Inv ention
[ 0003 ] The pr es ent inv ention r ela tes to gener a lly to com
pos itions a nd s olid or a l dos a ge f or m s conta ining a pha r m a
ceutica lly a ctiv e ingr edient a nd a n a lk yls a ccha r ide, which
enha nces the or a l bioa v a ila bility f or the a bs or ption of the
a ctiv e ingr edient.
[ 0004] 2. Ba ck gr ound Inf or m a tion
[ 0005] Ther a peutic a gents a r e of ten com bined with v a r ious
s ur f a cta nts . Y et, s ur f a cta nts a r e f r eq uently ir r ita ting to the
s k in a nd other tis s ues , including m ucos a l m em br a nes s uch a s
thos e f ound in the nos e, m outh, eye, v a gina , r ectum , es opha
gus , intes tina l tr a ct, a nd the lik e. Ma ny s ur f a cta nts a ls o ca us e
pr oteins to dena tur e, thus des tr oying their biologica l a ctiv ity.
Another s er ious lim ita tion to the dev elopm ent a nd us e of s uch
a gents is the a bility to deliv er them s a f ely, non- inv a s iv ely,
ef ? ciently a nd s ta bly to the s ite of a ction. Ther ef or e, a n idea l
enha ncing s ur f a cta nt will s ta biliz e the ther a peutic a gent, be
non- tox ic a nd non- ir r ita ble to the s k in or m ucos a l s ur f a ces ,
ha v e a ntiba cter ia l a ctiv ity, a nd enha nce the pa s s a ge or
a bs or ption of the ther a peutic a gent thr ough v a r ious m em
br a ne ba r r ier s without da m a ging the s tr uctur a l integr ity a nd
biologica l f unction of the m em br a ne a nd incr ea s e bioa v a il
a bility of the a gent.
[ 0006] In s pite of the m a ny a ttr a ctiv e a s pects of peptides
a nd pr oteins a s potentia l ther a peutic a gents , their s us ceptibil
Jun. 12, 2014
ity to dena tur a tion, hydr olys is , a nd poor a bs or ption in the
ga s tr ointes tina l tr a ct m a k es them uns uita ble f or or a l a dm in
is tr a tion, typica lly r eq uir ing a dm inis tr a tion by inj ection. This
r em a ins a m a j or s hor tcom ing. Com pa r ed to s m a ll m olecule
dr ugs , peptides a r e cons ider a bly les s s ta ble. Ca r ef ul a ttention
m us t be pa id to f or m ula tion a nd s tor a ge to a v oid unwa nted
degr a da tion. Som e pr oteins , pa r ticula r ly pr oteins with s ub
s ta ntia lly non- na tur a lly occur r ing a m ino a cid s eq uences ca n
be im m unogenic. Upon inj ection, im m une cells m a y be
r ecr uited to the s ite of inj ection a nd a hum or a l or cellula r
im m une r es pons e m a y be induced. Aggr ega ted peptides a r e
k nown to be m or e pr one to eliciting a n im m unogenic
r es pons e tha n m onom er s . This m a y be a v oided to a gr ea ter to
or les s er ex tent if the peptide ca n be dir ectly a bs or bed f r om
the ga s tr ointes tina l tr a ct into s ys tem ic cir cula tion. Ther ef or e,
while the r a nge of clinica l indica tions f or ther a peutic pr oteins
a nd peptides is q uite br oa d, the a ctua l num ber of s uch ther a
peutics in gener a l us e toda y is q uite s m a ll com pa r ed to the
num ber of chem ica lly s ynthes iz ed a nd or a lly a ctiv e pha r m a
ceutica ls cur r ently on the m a r k et. In r ecent yea r s , dev elop
m ent of a la r ge cla s s of a lk yls a ccha r ide deliv er y enha ncem ent
a gents , f or ex a m ple, m olecules tha t pr ov ide intr a na s a l bio
a v a ila bilities , com pa r a ble to thos e a chiev ed by inj ection ha v e
been inv es tiga ted. W hile r ecent dev elopm ents in intr a na s a l
deliv er y f or pr oteins a nd peptides a r e cr ea ting new a nd
ex pa nded oppor tunities f or pr a ctica l clinica l us es of peptides ,
pr oteins , a nd other m a cr om olecula r ther a peutics , f ew, if a ny,
peptides a ppea r to be a dm inis tr a ble or a lly due to una ccept
a bly low or a l bioa v a ila bility. A num ber of s tudies ha v e been
conducted to dem ons tr a te or a l bioa v a ila bility f or a v a r iety of
peptide dr ugs . Thes e s tudies us ed a v a r iety of a bs or ption
enha ncer s a s well a s phys ica l pr oces s es s uch a s m icr oniz a
tion. For ex a m ple a m ong f or m ula tions s peci? ca lly optim iz ed
f or or a l deliv er y, ins ulin ex hibited only 3 % or a l bioa v a ila bil
ity ( Ba dwin et a l. , 2009) . Ca lcitonin ex hibited only 05- 14%
or a l bioa v a ila bility ( Buck lin 2002) . Pa r a thyr oid hor m one ha s
been s hown to ex hibit 2. 1% or a l bioa v a ila bility ( Leone- Ba y
et a l. , 2001) . Ther e a r e two pr incipa l biochem ica l pr oblem s
lim iting the or a l a bs or ption of peptides . The ? r s t r ela tes to the
s us ceptibility of peptides to hydr olys is in the ga s tr ointes tina l
tr a ct. The s econd r ela tes to intr ins ica lly poor a bs or ption
a cr os s the intes tina l m ucos a l m em br a ne.
[ 0007 ] Incor por a tion of non- s ta nda r d a m ino a cids into pep
tide s eq uences ha s been s hown to r educe hydr olys is or s low
m eta bolis m f or s om e peptides . N on- s ta nda r d a m inoa cyl r es i
dues ha v e been incor por a ted into a num ber of dr ugs f or this
pur pos e a llowing the dr ugs to r em a in a ctiv e f or a longer
per iod of tim e tha n other wis e pos s ible. N on- s ta nda r d a m ino
a cids a r e thos e a m ino a cids tha t a r e not a m ong the 22 na tu
r a lly occur r ing L- a m ino a cids f ound in pr oteins . Ther e ex is t a
v a s t num ber of non- s ta nda r d a m ino a cids tha t m a y be con
s ider ed f or s uch us e in either the D or L con? gur a tion. A f ew
ex a m ples include, but a r e not lim ited to, a llylglycine, ( 2S, 3 R,
4S) - 0t- ( ca r box ycyclopr opyl) glycine, ( x - cyclohex ylglycine,
C- pr opa r gylglycine, ( x - neopentylglycine, ( x - cyclopr opylgly
cine, N - la ur oyls a r cos ine s odium s a lt, N - ( 4- hydr ox yphenyl)
glycine, N - ( 2- f ur oyl) glycine, na phthylglycine, phenylgly
cine, la nthionine, 2- a m inois obutyr ic a cid, dehydr oa la nine,
ga m m a - a m inobutyr ic a cid.
[ 0008 ] Som e s peci? c ex a m ples of non s ta nda r d a m ino
a cids us ed in dr ugs include D - 4- hydr ox yphenylglycine
which is incor por a ted into the a ntiba cter ia l dr ug Am ox icillin,
D - phenylglycine which is incor por a ted into the a ntihyper ten
US 2014/0162965 A1
s iv e dr ug Ena la pr il, a nd ( 2R, 3 S) - phenylis os er ine which is
incor por a ted into the a ntineopla s tic dr ug Ta x ol.
[ 0009] In the ca s e of peptide dr ugs , D - 2- N a phthyla la nine is
incor por a ted into the endom etr ios is dr ug N a f a r elin. The
D - is om er s of na tur a lly occur r ing L- a m ino a cids a r e f r e
q uently us ed to incr ea s e s ta bility of peptide dr ugs . Ex a m ples
of D - a m ino a cid s ta biliz ed peptides include the a nti- obes ity
peptide D - Leu- OB3 ( Lee et a l. , 2010) a nd the CCR5 a nti
H IV dr ug D - a la - peptide T ( D APTA) ( Ruf f et a l. , 2001)
a m ong other s .
[ 0010] Enz ym a tic hydr olys is in the ga s tr ointes tina l tr a ct
m a y a ls o be r educed or elim ina ted by a ddition of s peci? c
enz ym e inhibitor s s uch a s ba citr a cin, bes ta tin, a m a s ta tin,
bor oleucin, bor ov a line, a pr otinin, a nd tr yps in inhibitor
a m ong other s .
[ 0011] Alk yls a ccha r ides ha v e been dem ons tr a ted to
enha nce or a l a bs or ption of s m a ll m olecules , a nd peptides ,
when pr es ented a s a q ueous s olutions of the a lk yls a ccha r ide
a nd the s m a ll m olecule or peptide. In s uch s olutions , the
concentr a tion of the a lk yls a ccha r ide is higher tha n the cr itica l
m icelle concentr a tion ( CMC) . An ex a m ple includes the or a l
deliv er y of octr eotide in a q ueous s olution us ing dodecyl
beta - D - m a ltos ide a s the a lk yls a ccha r ide a bs or ption enha ncer .
Another ex a m ple is or a l deliv er y of a leptin- r ela ted s ynthetic
peptide ins ulin s ens itiz er us ing a n a q ueous dodecyl m a ltos ide
s olution. Y et a nother ex a m ple is or a l deliv er y of ex ena tide a nd
pr a m lintide us ing dodecyl- beta - D - m a ltos ide in a n a q ueous
s olution a s the a lk yls a ccha r ide a bs or ption enha ncer . Y et
a nother ex a m ple is or a l deliv er y of hepa r in us ing tetr a decyl
m a ltos ide in a n a q ueous s olution a s the a lk yl s a ccha r ide
a bs or ption enha ncer ( s ee f or ex a m ple, Ma ggio a nd Gr a s s o,
Regula tor y Peptides 167 ( 2011) 23 3 - 23 8 ; N ov a k ov ic, et a l. ,
Peptides , 43 ( 2013 ) 167 - 163 ; Leinung M C et a l. , Regula tor y
Peptides 17 9: 3 3 - 3 8 ( 2012) ; Y a ng, et a l. , ( 2005) Jour na l of
D r ug Ta r geting, 13 : 1, 29- 3 8 ) .
SUMMARY OF TH E IN VEN TION
[ 0012] The pr es ent inv ention is ba s ed, in pa r t, on the dev el
opm ent of a ther a peutic com pos ition conta ining a dr ug
enha ncing a gent us ef ul f or incr ea s ing the a bs or ption a nd
bioa v a ila bility of the dr ug, while a t the s a m e tim e a v oiding
v a r ious a dv er s e tox ic ef f ects of dr ug. In pa r ticula r , the dr ug
enha ncing a gents of the inv ention conta in a non- tox ic s ur f a c
ta nt cons is ting of a t lea s t a n a lk yl glycos ide a nd/or s a ccha r ide
a lk yl es ter . One a dv a nta ge of the ther a peutic com pos itions of
the inv ention is tha t they per m it a dm inis tr a tion a nd deliv er y
of the ther a peutic a gents with high bioa v a ila bilities a t con
centr a tions of enha ncing a gents tha t a r e dr a m a tica lly below
their s o- ca lled no obs er v a ble a dv er s e ef f ect lev els ( their
N OAEL s ) . Accor dingly, the pr es ent inv ention pr ov ides com
pos itions , including a lk yl glycos ides a nd/ or s a ccha r ide a lk yl
es ter s a nd a ther a peutic a gent ( e. g. s m a ll m olecule or ga nic
dr ug m olecules , low m olecula r weight peptides s uch a s
Ex ena tide, GLP- 1 a nd the lik e, pr oteins , a nd non- peptide
ther a peutic polym er s s uch a s low m olecula r weight hepa r in
a nd inhibitor y RN A) , m ethods of a dm inis ter ing a nd us ing the
com pos itions e. g. v ia the or a l, ocula r , na s a l, na s ola cr im a l,
inha la tion or pulm ona r y, or a l ca v ity ( s ublingua l or Bucca l
cell) or cer ebr a l s pina l ? uid ( CSF) deliv er y r oute, a nd m eth
ods of a m elior a ting a dis ea s e s ta te in a s ubj ect by a dm inis
tr a tion of s uch com pos itions .
[ 0013 ] Pr ev ious ly, no ex a m ples of the us e of a lk yls a ccha
r ides a s a bs or ption enha ncer s in s olid dos a ge f or m s s uch a s
ta blets ha s been dem ons tr a ted. W hile a lk yls a ccha r ides s uch
Jun. 12, 2014
a s dodecyl m a ltos ide, n- tetr a decyl m a ltos ide ha v e r ela tiv ely
high lev els of s olubility in a q ueous s olution, they dis s olv e
only v er y s lowly a nd beca us e a s igni? ca nt por tion of the
m olecules a r e com pr is ed of the hydr ophobic linea r a lk yl
cha ins . In or der to ens ur e com plete dis s olution of thes e a lk yl
s a ccha r ides in a q ueous s olution, the a q ueous s olution is
m ildly hea ted a nd the conta iner holding the a q ueous m edia
a nd the a lk yls a ccha r ide is either s tir r ed or a gita ted continu
ous ly f or up to 15 to 3 0 m in. in or der to ens ur e com plete
dis s olution of the a lk yls a ccha r ide. As a r es ult, it wa s not
ex pected tha t s olid dos a ge f or m s conta ining a n a lk yls a ccha
r ide a nd the dr ug s ubs ta nce would bene? t f r om the a bs or ption
enha ncing ef f ect of the a lk yls a ccha r ide s ince the s low dis s o
lution of the a lk yls a ccha r ide in the a q ueous ga s tr ointes tina l
contents would not be ex pected to pr oduce a s uf ? ciently high
concentr a tion or com pa r a ble concentr a tion to tha t a chiev ed in
the a q ueous f or m ula tions bef or e the dr ug s ubs ta nce a nd the
a lk yls a ccha r ide m olecules dif f us e a wa y f r om ea ch other
thr oughout the ga s tr ointes tina l contents diluting the r ela tiv e
concentr a tions of ea ch. Sur pr is ingly, s olid dos a ge f or m s
com pr is ing a n a lk yls a ccha r ide, a nd a pha r m a cologica lly
a ctiv e s ubs ta nce, a long with other ina ctiv e ex cipients f or m ed
into a ta blet wer e f ound to pr ov ide a s ubs ta ntia l incr ea s e in
or a l bioa v a ila bility of the pha r m a cologica lly a ctiv e s ub
s ta nce, a s s hown in the ex a m ples . In a ddition to a ctiv e pha r
m a ceutica l s ubs ta nce a nd a lk yls a ccha r ide a bs or ption
enha ncer , other ina ctiv e ex cipients m a y include by wa y of
ex a m ple ca ndelilla wa x , hypr om ellos e, m a gnes ium s tea r a te,
m icr ocr ys ta lline cellulos e, polyethylene glycol, pov idone,
a nd tita nium diox ide.
[ 0014] In one a s pect, the pr es ent inv ention r ela tes to a s ur
f a cta nt com pos ition ha v ing a t lea s t one a lk yl glycos ide a nd/or
a t lea s t one s a ccha r ide a lk yl es ter , a nd when a dm ix ed, m ix ed
or blended with a ther a peutic a gent, a dr ug, or biologica lly
a ctiv e com pound, the s ur f a cta nt s ta biliz es the biologica l
a ctiv ity a nd incr ea s es the bioa v a ila bility of the dr ug.
[ 0015] Accor dingly, in one a s pect, the inv ention pr ov ides a
ther a peutic com pos ition ha v ing a t lea s t one biologica lly
a ctiv e com pound a nd a t lea s t one s ur f a cta nt, wher ein the
s ur f a cta nt f ur ther cons is ts of a t lea s t one a lk yl glycos ide
a nd/or s a ccha r ide a lk yl es ter or s ucr os e es ter a nd wher ein the
ther a peutic com pos ition s ta biliz es the biologica lly a ctiv e
com pound f or a t lea s t a bout 6 m onths , or m or e, a nd f r om
a bout 4 C. to a bout 25 C.
[ 0016] The inv ention a ls o pr ov ides a m ethod of a dm inis
ter ing a ther a peutic com pos ition ha v ing a s ur f a cta nt includ
ing a t lea s t one a lk yl glycos ide a nd/or s a ccha r ide a lk yl es ter
a dm ix ed, m ix ed, or blended with a t lea s t one ther a peutic
a gent, or a dr ug, or biologica lly a ctiv e com pound, a nd a dm in
is ter ed or deliv er ed to a s ubj ect, wher ein the a lk yl ha s f r om
a bout 10 to 24, 10 to 20, 10 to 16, or 10 to 14 ca r bon a tom s ,
wher ein the s ur f a cta nt incr ea s es the s ta bility a nd bioa v a ila bil
ity of the ther a peutic a gent.
[ 0017 ] In yet a nother a s pect, the inv ention pr ov ides a
m ethod of incr ea s ing a bs or ption of a low m olecula r weight
com pound into the cir cula tor y s ys tem of a s ubj ect by a dm in
is ter ing the com pound v ia the or a l, ocula r , na s a l, na s ola cr i
m a l, inha la tion or pulm ona r y, or a l ca v ity ( s ublingua l or Buc
ca l cell) , or CSF deliv er y r oute when a dm ix ed, m ix ed or
blended with a n a bs or ption incr ea s ing a m ount of a s uita ble
s ur f a cta nt, wher ein the s ur f a cta nt is a nontox ic a nd nonionic
hydr ophobic a lk yl j oined by a link a ge to a hydr ophilic s a c
cha r ide. Such low m olecula r weight com pounds include but
a r e not lim ited to, nicotine, inter f er on, PY Y , GLP- l , s ynthetic
US 2014/0162965 A1
ex endin- 4, pa r a thyr oid hor m one, hum a n gr owth hor m one, or
a s m a ll or ga nic m olecule. Additiona l low m olecula r weight
com pounds include a ntis ens e oligonucleotides or inter f er ing
RN A m olecules ( e. g. , s iRN A or RN Ai) .
[ 0018 ] The pr es ent inv ention a ls o pr ov ides a m ethod of
tr ea ting dia betes including a dm inis ter ing to a s ubj ect in need
ther eof v ia the or a l, ocula r , na s a l, na s ola cr im a l, inha la tion or
pulm ona r y, or or a l ca v ity ( s ublingua l or Bucca l cell) , a blood
glucos e r educing a m ount of a ther a peutic com pos ition, f or
ex a m ple, a n incr etin m im etic a gent or a f unctiona l eq uiv a lent
ther eof , a nd a n a bs or ption incr ea s ing a m ount of a s uita ble
nontox ic, nonionic a lk yl glycos ide ha v ing a hydr ophobic
a lk yl gr oup j oined by a link a ge to a hydr ophilic s a ccha r ide,
ther eby incr ea s ing the a bs or ption of incr etin m im etic a gent or
ins ulin a nd lower ing the lev el of blood glucos e a nd tr ea ting
dia betes in the s ubj ect.
[ 0019] The pr es ent inv ention a ls o pr ov ides a m ethod of
tr ea ting conges tiv e hea r t f a ilur e in a s ubj ect including a dm in
is ter ing to the s ubj ect in need ther eof v ia the or a l, ocula r ,
na s a l, na s ola cr im a l, or inha la tion deliv er y r oute, a ther a peu
tica lly ef f ectiv e a m ount of a com pos ition com pr is ing a
GLP- l peptide or a f unctiona l eq uiv a lent ther eof , a nd a n
a bs or ption incr ea s ing a m ount of a s uita ble nontox ic, non
ionic a lk yl glycos ide ha v ing a hydr ophobic a lk yl j oined by a
link a ge to a hydr ophilic s a ccha r ide, ther eby tr ea ting the s ub
j ect.
[ 0020] In a nother a s pect, the inv ention pr ov ides a m ethod
of tr ea ting obes ity or dia betes a s s ocia ted with obes ity in a
s ubj ect com pr is ing a dm inis ter ing to a s ubj ect in need ther eof
v ia the or a l, ocula r , na s a l, na s ola cr im a l, inha la tion or CSF
deliv er y r oute, a ther a peutica lly ef f ectiv e a m ount of a com
pos ition com pr is ing a PY Y peptide or a f unctiona l eq uiv a lent
ther eof , a nd a n a bs or ption incr ea s ing a m ount of a s uita ble
nontox ic, nonionic a lk yl glycos ide ha v ing a hydr ophobic
a lk yl j oined by a link a ge to a hydr ophilic s a ccha r ide, ther eby
tr ea ting the s ubj ect.
[ 0021] In a nother a s pect, the inv ention pr ov ides a m ethod
of incr ea s ing a bs or ption of a low m olecula r weight ther a peu
tic com pound into the cir cula tor y s ys tem of a s ubj ect by
a dm inis ter ing v ia the or a l, ocula r , na s a l, na s ola cr im a l, inha
la tion or CSF deliv er y r oute the com pound a nd a n a bs or ption
incr ea s ing a m ount of a s uita ble nontox ic, nonionic a lk yl gly
cos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a link a ge
to a hydr ophilic s a ccha r ide, wher ein the com pound is f r om
a bout 1- 3 0 k D , with the pr ov is o tha t the com pound is not
ins ulin, ca lcitonin, or gluca gon when the r oute of a dm inis tr a
tion is or a l, ocula r , na s a l, or na s ola cr im a l.
[ 0022] The pr es ent inv ention a ls o pr ov ides a m ethod of
incr ea s ing a bs or ption of a low m olecula r weight ther a peutic
com pound into the cir cula tor y s ys tem of a s ubj ect by a dm in
is ter ing v ia the or a l, ocula r , na s a l, na s ola cr im a l, inha la tion or
pulm ona r y, or a l ca v ity ( s ublingua l or Bucca l cell) or CSF
deliv er y r oute the com pound a nd a n a bs or ption incr ea s ing
a m ount of a s uita ble nontox ic, nonionic a lk yl glycos ide ha v
ing a hydr ophobic a lk yl j oined by a link a ge to a hydr ophilic
s a ccha r ide, wher ein the com pound is f r om a bout 1- 3 0 k ilo
D a ltons ( k D ) , with the pr ov is o tha t the s ubj ect does not ha v e
dia betes when deliv er y is v ia the or a l, ocula r , na s a l or na s o
la cr im a l r outes .
[ 0023 ] In one a s pect of the inv ention, ther e is pr ov ided a
pha r m a ceutica l com pos ition ha v ing a s uita ble nontox ic, non
ionic a lk yl glycos ide ha v ing a hydr ophobic a lk yl gr oup
j oined by a link a ge to a hydr ophilic s a ccha r ide in com bina
Jun. 12, 2014
tion with a ther a peutica lly ef f ectiv e a m ount of Ex ena tide
( ex endin- 4) in a pha r m a ceutica lly a ccepta ble ca r r ier .
[ 0024] In one a s pect, the inv ention pr ov ides a pha r m a ceu
tica l com pos ition ha v ing a s uita ble nontox ic, nonionic a lk yl
glycos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a link
a ge to a hydr ophilic s a ccha r ide in com bina tion with a ther a
peutica lly ef f ectiv e a m ount of GLP- 1 in a pha r m a ceutica lly
a ccepta ble ca r r ier .
[ 0025] In one a s pect, the inv ention pr ov ides a pha r m a ceu
tica l com pos ition ha v ing a s uita ble nontox ic, nonionic a lk yl
glycos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a link
a ge to a hydr ophilic s a ccha r ide in com bina tion with a ther a
peutica lly ef f ectiv e a m ount of nicotine in a pha r m a ceutica lly
a ccepta ble ca r r ier .
[ 0026] In one a s pect, the inv ention pr ov ides a pha r m a ceu
tica l com pos ition com pr is ing a s uita ble nontox ic, nonionic
a lk yl glycos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a
link a ge to a hydr ophilic s a ccha r ide in com bina tion with a
ther a peutica lly ef f ectiv e a m ount of inter f er on in a pha r m a
ceutica lly a ccepta ble ca r r ier .
[ 0027 ] In one a s pect, the inv ention pr ov ides pha r m a ceuti
ca l com pos ition ha v ing a s uita ble nontox ic, nonionic a lk yl
glycos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a link
a ge to a hydr ophilic s a ccha r ide in com bina tion with a ther a
peutica lly ef f ectiv e a m ount of PY Y in a pha r m a ceutica lly
a ccepta ble ca r r ier .
[ 0028 ] In one a s pect, the inv ention pr ov ides a pha r m a ceu
tica l com pos ition ha v ing a s uita ble nontox ic, nonionic a lk yl
glycos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a link
a ge to a hydr ophilic s a ccha r ide in com bina tion with a ther a
peutica lly ef f ectiv e a m ount of pa r a thyr oid hor m one in a pha r
m a ceutica lly a ccepta ble ca r r ier .
[ 0029] In one a s pect, the inv ention pr ov ides a pha r m a ceu
tica l com pos ition ha v ing a s uita ble nontox ic, nonionic a lk yl
glycos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a link
a ge to a hydr ophilic s a ccha r ide in com bina tion with a ther a
peutica lly ef f ectiv e a m ount of a peptide ha v ing a m olecula r
weight of a bout 1- 7 5 k D in a pha r m a ceutica lly a ccepta ble
ca r r ier , with the pr ov is o tha t the peptide is not ins ulin, ca lci
tonin, a nd gluca gon.
[ 003 0] In one a s pect, the inv ention pr ov ides a pha r m a ceu
tica l com pos ition ha v ing a s uita ble nontox ic, nonionic a lk yl
glycos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a link
a ge to a hydr ophilic s a ccha r ide in com bina tion with a ther a
peutica lly ef f ectiv e a m ount er ythr opoietin in a pha r m a ceuti
ca lly a ccepta ble ca r r ier .
[ 003 1] In one a s pect, the inv ention pr ov ides a pha r m a ceu
tica l com pos ition ha v ing a ther a peutica lly ef f ectiv e a m ount
of a n oligonucleotide in com bina tion with a n a bs or ption
incr ea s ing a m ount of a n a lk ylglycos ide. The oligonucleotide
ca n be a n a ntis ens e oligonucleotide or inter f er ing RN A m ol
ecules , s uch a s s iRN A or RN Ai. The oligonucleotide typi
ca lly ha s a m olecula r weight of a bout 1- 20 k D a nd is f r om
a bout 1- 100, 1- 50, 1- 3 0, 1- 25 or 15- 25 nucleotides in length.
In a nother a s pect, the oligonucleotide ha s a m olecula r weight
of a bout 5- 10 k D . In one a s pect, the a lk ylglycos ide is tetr a de
cyl- beta - D - m a ltos ide.
[ 003 2] In yet a nother a s pect, the inv ention pr ov ides a
m ethod of incr ea s ing the bioa v a ila bility of a low m olecula r
weight oligonucleotide in a s ubj ect by a dm inis ter ing the com
pound with a n a bs or ption incr ea s ing a m ount of a n a lk ylgly
cos ide, ther eby incr ea s ing the bioa v a ila bility of the com
pound in the s ubj ect. In one a s pect, the a lk ylglycos ide is
tetr a decyl- beta - D - m a ltos ide.
US 2014/0162965 A1
[ 003 3 ] In one a s pect, the inv ention pr ov ides a m ethod of
incr ea s ing a bs or ption of a com pound into the CSF of a s ubj ect
ha v ing a dm inis ter ed intr a na s a lly the com pound a nd a n
a bs or ption incr ea s ing a m ount of a s uita ble nontox ic, non
ionic a lk yl glycos ide ha v ing a hydr ophobic a lk yl gr oup
j oined by a link a ge to a hydr ophilic s a ccha r ide.
[ 003 4] In yet a nother a s pect, the inv ention pr ov ides a pha r
m a ceutica l com pos ition ha v ing a s uita ble nontox ic, nonionic
a lk yl glycos ide ha v ing a hydr ophobic a lk yl gr oup j oined by a
link a ge to a hydr ophilic s a ccha r ide in com bina tion W ith a
m ucos a l deliv er y- enha ncing a gent s elected f r om :
[ 003 5] ( a ) a n a ggr ega tion inhibitor y a gent;
[ 003 6] ( b) a cha r ge- m odif ying a gent;
[ 003 7 ] ( c) a pH contr ol a gent;
[ 003 8 ] ( d) a degr a da tiv e enz ym e inhibitor y a gent;
[ 003 9] ( e) a m ucolytic or m ucus clea r ing a gent;
[ 0040] ( f ) a cilios ta tic a gent;
[ 0041] ( g) a m em br a ne penetr a tion- enha ncing a gent
s elected f r om :
[ 0042] ( i) a s ur f a cta nt; ( ii) a bile s a lt; ( ii) a phos pholipid
a dditiv e, m ix ed m icelle, lipos om e, or ca r r ier ; ( iii) a n
a lcohol; ( iv ) a n ena m ine; ( v ) a n N O donor com pound;
( v i) a long- cha in a m phipa thic m olecule; ( v ii) a s m a ll
hydr ophobic penetr a tion enha ncer ; ( v iii) s odium or a
s a licylic a cid der iv a tiv e; ( ix ) a glycer ol es ter of
a cetoa cetic a cid; ( x ) a cyclodex tr in or beta - cyclodex tr in
der iv a tiv e; ( x i) a m edium - cha in f a tty a cid; ( x ii) a chela t
ing a gent; ( x iii) a n a m ino a cid or s a lt ther eof ; ( x iv ) a n
N - a cetyla m ino a cid or s a lt ther eof ; ( x v ) a n enz ym e deg
r a da tiv e to a s elected m em br a ne com ponent; ( ix ) a n
inhibitor of f a tty a cid s ynthes is ; ( x ) a n inhibitor of cho
les ter ol s ynthes is ; a nd ( x i) a ny com bina tion of the m em
br a ne penetr a tion enha ncing a gents r ecited in ( i) - ( x ) ;
[ 0043 ] ( h) a m odula tor y a gent of epithelia l j unction phys i
Ology;
[ 0044] ( i) a v a s odila tor a gent;
[ 0045] ( j ) a s electiv e tr a ns por t- enha ncing a gent; a nd
[ 0046] ( k ) a s ta biliz ing deliv er y v ehicle, ca r r ier , m ucoa d
hes iv e, s uppor t or com plex - f or m ing s pecies W ith W hich the
com pound is ef f ectiv ely com bined, a s s ocia ted, conta ined,
enca ps ula ted or bound r es ulting in s ta biliz a tion of the com
pound f or enha nced na s a l m ucos a l deliv er y, W her ein the f or
m ula tion of the com pound W ith the intr a na s a l deliv er y- en
ha ncing a gents pr ov ides f or incr ea s ed bioa v a ila bility of the
com pound in a blood pla s m a of a s ubj ect.
[ 0047 ] In one a s pect, the inv ention pr ov ides a m ethod of
incr ea s ing a bs or ption of a low m olecula r weight com pound
into the cir cula tor y s ys tem of a s ubj ect by a dm inis ter ing, v ia
the or a l, ocula r , na s a l, na s ola cr im a l, inha la tion or pulm ona r y,
or a l ca v ity ( s ublingua l or Bucca l cell) or CSF deliv er y r oute
( a ) the com pound; ( b) a n a bs or ption incr ea s ing a m ount of a
s uita ble nontox ic, nonionic a lk yl glycos ide ha v ing a hydr o
phobic a lk yl gr oup j oined by a link a ge to a hydr ophilic s a c
cha r ide; a nd ( c) a m ucos a l deliv er y- enha ncing a gent.
[ 0048 ] In one a s pect, the inv ention pr ov ides a m ethod of
contr olling ca lor ic inta k e by a dm inis ter ing a com pos ition
ha v ing a ther a peutic ef f ectiv e a m ount of ex endin- 4, or r ela ted
GLP- l peptide, W ith a n ef f ectiv e a m ount of Intr a v a il a lk yl
s a ccha r ide.
[ 0049] In a nother a s pect, the inv ention pr ov ides a m ethod
of contr olling blood glucos e lev els in a s ubj ect by a dm inis
ter ing to a s ubj ect a com pos ition com pr is ing a ther a peutic
ef f ectiv e a m ount of ex endin- 4, or r ela ted GLP- l peptide, W ith
a n ef f ectiv e a m ount of Intr a v a il a lk yl s a ccha r ide.
Jun. 12, 2014
[ 0050] Still, in a nother a s pect, the inv ention pr ov ides a
contr olled r elea s e dos a ge com pos ition com pr is ing:
[ 0051] ( a ) a cor e com pr is ing:
[ 0052] ( i) a t lea s t one ther a peutic a gent or dr ug;
[ 0053 ] ( ii) a t lea s t one a lk yl glycos ide a nd/or s a ccha
r ide a lk yl es ter ; a nd
[ 0054] ( b) a t lea s t one m em br a ne coa ting s ur r ounding
the cor e, W her ein the coa ting is im per m ea ble, per m e
a ble, s em i- per m ea ble or por ous a nd becom es m or e per
m ea ble upon s us ta ined conta ct W ith contents of the ga s
tr ointes tina l tr a ct.
[ 0055] In a nother em bodim ent, the inv ention pr ov ides a
m ethod of a dm inis ter ing a n a lk ylglycos ide com pos ition by
a dm inis ter ing a ther a peutica lly ef f ectiv e a m ount of a t lea s t
one a lk yglycos ide ha v ing a n a lk yl cha in length f r om a bout 12
to a bout 14 ca r bon a tom s , a t lea s t one s a ccha r ide W ith a n
a ntiba cter ia l a ctiv ity, a nd a t lea s t one ther a peutic a gent.
[ 0056] Still in a nother em bodim ent, the inv ention pr ov ides
a com pos ition ha v ing a t lea s t one dr ug s elected f r om the
gr oup cons is ting of ins ulin, PY Y , Ex endin- 4 or other GLP- l
r ela ted peptide, hum a n gr owth hor m one, ca lcitonin, pa r a thy
r oid hor m one, tr unca ted pa r a thyr oid hor m one peptides s uch
a s PTH 1- 3 4, EPO, inter f er on a lpha , inter f er on beta , inter
f er on ga m m a , a nd GCSF a nd a t lea s t one a lk yl s a ccha r ide
ha v ing a ntiba cter ia l a ctiv ity.
[ 0057 ] In one a s pect, the inv ention pr ov ides a n a ntiba cter ia l
a lk yl s a ccha r ide com pos ition, W hich includes n- D odecyl- 4
O- ( x - D - glucopyr a nos yl- [ 3 - D - glucopyr a nos ide or n- tetr a de
cyl- 4- O- ( x - D - glucopyr a nos yl- [ 3 - D - glucopyr a nos ide.
[ 0058 ] Y et, in a nother a s pect, the inv ention pr ov ides a n
a q ueous dr ug com pos ition f or tr a ns m ucoca l or tr a ns der m a l
a dm inis tr a tion ha v ing a t lea s t one dr ug a nd a t lea s t one a nti
ba cter ia l a gent in a concentr a tion f r om a bout 0. 05% to a bout
0. 5% .
[ 0059] In a nother a s pect, the inv ention pr ov ides a f a s t- dis
per s ing dr ug f or m ula tion conta ining a m a tr ix m a ter ia l a nd a n
a lk yls a ccha r ide. The f or m ula tion m a y ha v e a Tm a x s ubs ta n
tia lly les s tha n, a nd a ? r s t- pa s s ef f ect s ubs ta ntia lly les s tha n
tha t obs er v ed f or a n eq uiv a lent f or m ula tion not conta ining a n
a lk yls a ccha r ide. In one em bodim ent, the f or m ula tion m a y
conta in a bout 0. 1% to 10% a lk yls a ccha r ide, a nd ex hibits a
Tm a x s ubs ta ntia lly les s tha n s ix hour s a nd a ? r s t- pa s s ef f ect
of les s tha n 40% . The a lk ylglycos ide m a y be a ny s uita ble
a lyk ylglycos ide a nd in a pr ef er r ed a s pect is dodecyl m a lto
s ide, tetr a decyl m a ltos ide, s ucr os e dodeca noa te, or s ucr os e
m ono- a nd di- s tea r a te. The f or m ula tion m a y include a v a r iety
of dif f er ent ther a peutics , s uch a s but not lim ited to m ela tonin,
r a lox if ene, ola nz a pene a nd diphenhydr a m ine.
[ 0060] In a nother a s pect, the inv ention pr ov ides a m ethod
f or pr ov iding a n ex tended a bs or ption cur v e by a ttenua ting the
a lk yls a ccha r ide concentr a tion in dr ug f or m ula tion to ba la nce
ga s tr ic a nd bucca l deliv er y. For ex a m ple, this is per f or m ed by
pr ov iding a dr ug f or m ula tion including a m a tr ix m a ter ia l a nd
a n a lk yls a ccha r ide ha v ing a Tm a x s ubs ta ntia lly les s tha n, a nd
a ? r s t- pa s s ef f ect s ubs ta ntia lly les s tha n tha t obs er v ed f or a n
eq uiv a lent f or m ula tion not conta ining a n a lk yls a ccha r ide.
[ 0061] In one a s pect, the inv ention pr ov ides a pha r m a ceu
tica l com pos ition ha v ing a ther a peutica lly ef f ectiv e a m ount
of a bis pho s phona te a na log or a tr ipta n a na log in com bina tion
W ith a n a bs or ption incr ea s ing a m ount of a n a lk ylglycos ide. In
v a r ious em bodim ents , the bis phos phona te a na log m a y be
etidr ona te, clodr ona te, tiludr ona te, pa m idr ona te, ner idr ona te,
olpa dr ona te, a lendr ona te, iba ndr ona te, r is edr ona te, z oledr
ona te, a nd/ or pha r m a ceutica lly a ccepta ble a na logs ther eof . In
US 2014/0162965 A1
a n ex em pla r y em bodim ent, the bis phos phona te a na log is
a lendr ona te or pha r m a ceutica lly a ccepta ble a na log ther eof .
In v a r ious em bodim ents , the tr ipta n a na log m a y be s um a tr ip
ta n, r iz a tr ipta n, na r a tr ipta n, z olm itr ipta n, eletr ipta n, a lm ot
r ipta n, f r ov a tr ipta n a nd/ or pha r m a ceutica lly a ccepta ble a na
logs ther eof . In a n ex em pla r y em bodim ent, the tr ipta n a na log
is s um a tr ipta n or pha r m a ceutica lly a ccepta ble a na log ther eof .
In v a r ious em bodim ents , the a lk ylglycos ide is tetr a decyl
beta - D - m a ltos ide.
[ 0062] In yet a nother a s pect, the inv ention pr ov ides a
m ethod of incr ea s ing the bioa v a ila bility of a bis phos phona te
a na log or a tr ipta n a na log in a s ubj ect by a dm inis ter ing the
com pound with a n a bs or ption incr ea s ing a m ount of a n a lk y
lglycos ide, ther eby incr ea s ing the bioa v a ila bility of the com
pound in the s ubj ect.
[ 0063 ] In s till a nother a s pect, the inv ention pr ov ides a com
pos ition including a peptide, wher ein the peptide includes a
D - a m ino a cid or a s ite f or cycliz a tion, or com bina tion ther eof ,
a nd a t lea s t one a lk yls a ccha r ide, wher ein the a lk yls a ccha r ide
pr ov ides incr ea s ed enter a l a bs or ption of the peptide.
[ 0064] In yet a nother a s pect, the inv ention pr ov ides m ethod
of incr ea s ing enter a l a ds or ption of a peptide in a bipha s ic
m a nner . The m ethod includes or a lly or na s a lly a dm inis ter ing
to a s ubj ect a com pos ition com pr is ing a t lea s t one peptide,
wher ein the peptide com pr is es a D - a m ino a cid or a s ite f or
cycliz a tion, or com bina tion ther eof , a nd a t lea s t one a lk yls a c
cha r ide, wher ein the enter a l a bs or ption of the peptide is
incr ea s ed a nd s ys tem ic s er um lev els of the peptide a r e
incr ea s ed in a bipha s ic m a nner .
[ 0065] In yet a nother a s pect, the inv ention pr ov ides a
m ethod of incr ea s ing the bioa v a ila bility of a gluca gon- lik e
peptide- 1 ( GLP- l) a na log in a s ubj ect. The m ethod includes
a dm inis ter ing the a na log with a n a bs or ption incr ea s ing
a m ount of a n a lk ylglycos ide, ther eby incr ea s ing the bioa v a il
a bility of the a na log in the s ubj ect.
[ 0066] In yet a nother a s pect, the inv ention pr ov ides a pha r
m a ceutica l com pos ition including a gluca gon- lik e peptide- 1
( GLP- l) a na log; a nd a n a bs or ption incr ea s ing a m ount of a n
a lk ylglycos ide.
BRIEF D ESCRIPTION OF TH E D RAW IN GS
[ 0067 ] FIG. 1 is a gr a ph s howing the intr a na s a l per cent
bioa v a ila bility com pa r ed to intr a v enous inj ection a nd the
s ubj ect- to- s ubj ect coef ? cients of v a r ia tion f or MIACAL
CIN ( s a lm on ca lcitonin) with a nd without a lk yl glycos ide.
[ 0068 ] FIG. 2 is a gr a ph s howing the ef f ect of intr a na s a l
a dm inis tr a tion of ins ulin/0. 25% TD M ( ? lled cir cles ) a nd
intr a na s a l a dm inis tr a tion of ins ulin a lone ( open cir cles ) in
r educing blood glucos e lev els .
[ 0069] FIG. 3 is a gr a ph s howing the ef f ect of intr a na s a l
( clos ed tr ia ngles ) a nd intr a per itonea l ( IP) inj ection ( clos ed
cir cles ) a dm inis tr a tion of ex endin- 4/0. 25% TD M a nd IP
inj ection of s a line a lone, m inus TD M ( open cir cles ) in r educ
ing blood glucos e lev els f ollowing intr a per itonea l ( IP) inj ec
tion of glucos e ( i. e. , in a s o- ca lled glucos e toler a nce tes t ) .
[ 007 0] FIG. 4 is a gr a ph s howing the upta k e of 1 m g m ous e
p- Leu- 4] OB3 in 0. 3 % a lk ylglycos ide tetr a decyl- beta - D - m a l
tos ide ( Intia v a ilTM A3 ) by m a le Swis s W ebs ter Mice f ollow
ing a dm inis tr a tion by ga v a ge.
[ 007 1] FIG. 5 is a gr a ph s howing the upta k e of s um a tr ipta n
in 0. 5% a lk ylglycos ide tetr a decyl- beta - D - m a ltos ide ( Intr a
v a ilTM A3 ) by ca nines f or both or a l a nd r ecta l a dm inis tr a tion.
Jun. 12, 2014
[ 007 2] FIG. 6 is a gr a ph s howing the upta k e pr o? le of 3 0 pg
octr eotide in s odium a ceta te buf f er a f ter s ubcuta neous deliv
er y to m a le Swis s W ebs ter m ice.
[ 007 3 ] FIG. 7 is a gr a ph s howing the upta k e pr o? le of 3 0 pg
octr eotide in 0. 5% Intr a v a ilTM a f ter or a l deliv er y to m a le
Swis s W ebs ter m ice.
[ 007 4] FIG. 8 is a gr a ph s howing the upta k e pr o? le of 3 0 pg
octr eotide in 1. 5% Intr a v a ilTM a f ter or a l deliv er y to m a le
Swis s W ebs ter m ice.
[ 007 5] FIG. 9 is a gr a ph s howing the upta k e pr o? le of 3 0 pg
octr eotide in 3 . 0% Intr a v a ilTM a f ter or a l deliv er y to m a le
Swis s W ebs ter m ice.
[ 007 6] FIG. 10 is a gr a ph s howing blood glucos e lev els
a f ter or a l a dm inis tr a tion of a n a lk ylglycos ide com pos ition
including lir a glutide a nd cha llenge with glucos e.
[ 007 7 ] FIG. 11 is a gr a ph dis pla ying a dos e r es pons e cur v e.
[ 007 8 ] FIG. 12 is a gr a ph s howing the per cent of PE in the
pla s m a ( ca nine m odel) ov er tim e when deliv er ed with a lk y
lglycos ide, n- dodecyl- beta - D - m a ltos ide.
[ 007 9] FIG. 13 is gr a ph s howing the per cent of PE in the
pla s m a ( ca nine m odel) ov er tim e when deliv er ed with the
a lk ylglycos ide, s ucr os e m onododeca noa te.
D ETAILED D ESCRIPTION OF TH E IN VEN TION
[ 008 0] The pr es ent inv ention m a y be under s tood m or e
r ea dily by r ef er ence to the f ollowing deta iled des cr iption of
s peci? c em bodim ents a nd the Ex a m ples included ther ein.
[ 008 1] The pr es ent inv ention is ba s ed on the dis cov er y tha t
ther a peutic com pos itions com pr is ing of lea s t one dr ug a nd a t
lea s t one s ur f a cta nt, wher ein the s ur f a cta nt is com pr is ed of a t
lea s t one a lk yl glycos ide a nd/or a t lea s t one s a ccha r ide a lk yl
es ter a r e s ta ble, non- tox ic, non- ir r ita ting, a nti- ba cter ia l com
pos itions tha t incr ea s e bioa v a ila bility of the dr ug a nd ha v e no
obs er v a ble a dv er s e ef f ects when a dm inis ter ed to a s ubj ect.
[ 008 2] A ther a peutic com pos ition ca n cons is t of a n
a dm ix tur e with a n or ga nic or inor ga nic ca r r ier or ex cipient,
a nd ca n be com pounded, f or ex a m ple, with the us ua l non
tox ic, pha r m a ceutica lly a ccepta ble ca r r ier s f or ta blets , pel
lets , ca ps ules , s uppos itor ies , s olutions , em uls ions , s us pen
s ions , or other f or m s uita ble f or us e. The ca r r ier s , in a ddition
to thos e dis clos ed a bov e, ca n include glucos e, la ctos e, m a n
nos e, gum a ca cia , gela tin, m a nnitol, s ta r ch pa s te, m a gnes ium
tr is ilica te, ta lc, cor n s ta r ch, k er a tin, colloida l s ilica , pota to
s ta r ch, ur ea , m edium cha in length tr iglycer ides , dex tr a ns , a nd
other ca r r ier s s uita ble f or us e in m a nuf a ctur ing pr epa r a tions ,
in s olid, s em is olid, or liq uid f or m . In a ddition, a ux ilia r y s ta
biliZ ing, thick ening or color ing a gents ca n be us ed, f or
ex a m ple a s ta biliZ ing dr y a gent s uch a s tr iulos e.
[ 008 3 ] A dr ug is a ny ther a peutic com pound, or m olecule,
or ther a peutic a gent, or biologica lly a ctiv e com pound, includ
ing but not lim ited to nucleic a cids , s m a ll m olecules , pr oteins ,
polypeptides or peptides a nd the lik e.
[ 008 4] The ter m nucleic a cids or oligonucleotide a ls o
denotes D N A, cD N A, RN A, s iRN A, RN Ai, ds RN A a nd the
lik e, which encode tr a ns la ted a nd untr a ns la ted r egions or
inhibits tr a ns la ted or untr a ns la ted r egions of s tr uctur a l genes
encoding a peptide or pr otein or r egula tor y r egion. For
ex a m ple, a nucleic a cid of the inv ention ca n include 5' a nd 3 '
untr a ns la ted r egula tor y nucleotide s eq uences a s well a s tr a ns
la ted s eq uences a s s ocia ted with a s tr uctur a l gene. The ter m
nucleic a cids or oligonucleotide or gr a m m a tica l eq uiv a
lents a s us ed her ein, r ef er s to a t lea s t two nucleotides
cov a lently link ed together .
US 2014/0162965 A1
[ 008 5] Additiona lly, the ter m oligonucleotide r ef er s to
s tr uctur es including m odi? ed por tions s uch a s m odi? ed s uga r
m oieties , m odi? ed ba s e m oieties or m odi? ed s uga r link ing
m oieties . Thes e m odi? ed por tions f unction in a m a nner s im i
la r to na tur a l ba s es , na tur a l s uga r s a nd na tur a l pho s phodies ter
link a ges . Accor dingly, oligonucleotides m a y ha v e a lter ed
ba s e m oieties , a lter ed s uga r m oieties or a lter ed inter - s uga r
link a ges . Modi? ed link a ges m a y be, f or ex a m ple, phos phor a
m ide, phos phor othioa te, phos phor odithioa te, m ethyl phos
phona te, phos photr ies ter , phos phor a m ida te, O- m ethylphos
phor oa m idite link a ges , or peptide nucleic a cid ba ck bones a nd
link a ges . Other a na logs m a y include oligonucleotides with
pos itiv e ba ck bones , non- ionic ba ck bones a nd non- r ibos e
ba ck bones . The nucleic a cid m a y be D N A, both genom ic a nd
cD N A, RN A or a hybr id, wher e the nucleic a cid conta ins a ny
com bina tion of deox yr ibo- a nd r ibo- nucleotides , a nd a ny
com bina tion of na tur a l or m odi? ed ba s es , including ur a cil,
a denine, thym ine, cytos ine, gua nine, inos ine, x a tha nine,
hypox a tha nine, is ocytos ine, is ogua nine, ha logenta ted ba s es
a nd the lik e. Other m odi? ca tions m a y include, f or ex a m ple,
dea z a or a z a pur ines a nd pyr im idines us ed in pla ce of na tur a l
pur ine a nd pyr im idine ba s es ; pyr im idine ba s es ha v ing s ub
s tituent gr oups a t the 5- or 6- pos itions , pur ine ba s es ha v ing
a lter ed or r epla cem ent s ubs tituent gr oups a t the 2- , 6- or
8 - pos itions , or s uga r s ha v ing s ubs tituent gr oups a t their 2' - po
s ition, s ubs titutions f or one or m or e of the hydr ogen a tom s of
the s uga r , or ca r bocyclic or a cyclic s uga r s .
[ 008 6] The ter m a ntis ens e, a s us ed her ein, r ef er s to a ny
com pos ition conta ining a nucleic a cid s eq uence which is
com plem enta r y to a s peci? c nucleic a cid s eq uence. The ter m
a ntis ens e s tr a n is us ed in r ef er ence to a nucleic a cid s tr a nd
tha t is com plem enta r y to the s ens e s tr a nd. Antis ens e m ol
ecules m a y be pr oduced by a ny m ethod including s ynthes is or
tr a ns cr iption. Once intr oduced into a cell, the com plem enta r y
nucleotides com bine with na tur a l s eq uences pr oduced by the
cell to f or m duplex es a nd to block either tr a ns cr iption or
tr a ns la tion.
[ 008 7 ] Antis ens e m olecules include oligonucleotides com
pr is ing a s inge- s tr a nded nucleic a cid s eq uence ( either RN A or
D N A) ca pa ble of binding to ta r get r eceptor or liga nd m RN A
( s ens e) or D N A ( a ntis ens e) s eq uences . The a bility to der iv e
a n a ntis ens e or a s ens e oligonucleotide, ba s ed upon a cD N A
s eq uence encoding a giv en pr otein. Antis ens e or s ens e oligo
nucleotides f ur ther com pr is e oligonucleotides ha v ing m odi
? ed s uga r - phos phodies ter ba ck bones a nd wher ein s uch s uga r
link a ges a r e r es is ta nt to endogenous nuclea s es . Such oligo
nucleotides with r es is ta nt s uga r link a ges a r e s ta ble in v iv o
( i. e. , ca pa ble of r es is ting enz ym a tic degr a da tion) but r eta in
s eq uence s peci? city to be a ble to bind to ta r get nucleotide
s eq uences .
[ 008 8 ] RN Ai is a phenom enon in which the intr oduction of
ds RN A into a div er s e r a nge of or ga nis m s a nd cell types
ca us es degr a da tion of the com plem enta r y m RN A. In the cell,
long ds RN As a r e clea v ed into s hor t ( e. g. , 21- 25 nucleotide)
s m a ll inter f er ing RN As ( s iRN As ) , by a r ibonuclea s e. The
s iRN As s ubs eq uently a s s em ble with pr otein com ponents into
a n RN A- induced s ilencing com plex ( RISC) , unwinding in
the pr oces s . The a ctiv a ted RISC then binds to com plem enta r y
tr a ns cr ipts by ba s e pa ir ing inter a ctions between the s iRN A
a ntis ens e s tr a nd a nd the m RN A. The bound m RN A is then
clea v ed a nd s eq uence s peci? c degr a da tion of m RN A r es ults
in gene s ilencing. As us ed her ein, s ilencing r ef er s to a
m echa nis m by which cells s hut down la r ge s ections of chr o
m os om a l D N A r es ulting in s uppr es s ing the ex pr es s ion of a
Jun. 12, 2014
pa r ticula r gene. The RN Ai m a chiner y a ppea r s to ha v e
ev olv ed to pr otect the genom e f r om endogenous tr a ns pos a ble
elem ents a nd f r om v ir a l inf ections . Thus , RN Ai ca n be
induced by intr oducing nucleic a cid m olecules com plem en
ta r y to the ta r get m RN A to be degr a ded.
[ 008 9] Other ex a m ples of s ens e or a ntis ens e oligonucle
otides include thos e oligonucleotides which a r e cov a lently
link ed to or ga nic m oieties a nd other m oieties tha t incr ea s e
a f ? nity of the oligonucleotide f or a ta r get nucleic a cid
s eq uence, s uch a s poly- ( L- lys ine) . Fur ther s till, inter ca la ting
a gents , s uch a s ellipticine, a nd a lk yla ting a gents or m eta l
com plex es m a y be a tta ched to s ens e or a ntis ens e oligonucle
otides to m odif y binding s peci? cities of the a ntis ens e or s ens e
oligonucleotide f or the ta r get nucleotide s eq uence.
[ 0090] A peptide of the inv ention m a y be a ny m edica lly or
dia gnos tica lly us ef ul peptide or pr otein of s m a ll to m edium
s iz e ( i. e. up to a bout 15 k D , 3 0 k D , 40 k D , 50 k D , 60 k D , 7 0
k D , 8 0 k D , 90 k D , 100 k D , f or ex a m ple) . The m echa nis m s of
im pr ov ed polypeptide a bs or ption a r e des cr ibed in Us . Pa t.
N o. 5, 661, 13 0 which is her eby incor por a ted by r ef er ence in
its entir ety. Inv ention com pos itions ca n be m ix ed with a ll
s uch peptides , a lthough the degr ee to which the peptide ben
e? ts a r e im pr ov ed m a y v a r y a ccor ding to the m olecula r
weight a nd the phys ica l a nd chem ica l pr oper ties of the pep
tide, a nd the pa r ticula r s ur f a cta nt us ed. Ex a m ples of polypep
tides include v a s opr es s in, v a s opr es s in polypeptide a na logs ,
des m opr es s in, gluca gon, cor ticotr opin ( ACTH ) , gona dotr o
pin, ca lcitonin, C- peptide of ins ulin, pa r a thyr oid hor m one
( PTH ) , gr owth hor m one ( H G) , hum a n gr owth hor m one
( hGH ) , gr owth hor m one r elea s ing hor m one ( GH RH ) , ox yto
cin, cor ticotr opin r elea s ing hor m one ( CRH ) , s om a tos ta tin or
s om a tos ta tin polypeptide a na logs , gona dotr opin a gonis t or
gona dotr ophin a gonis t polypeptide a na logs , hum a n a tr ia l
na tr iur etic peptide ( AN P) , hum a n thyr ox ine r elea s ing hor
m one ( TRH ) , f ollicle s tim ula ting hor m one ( FSH ) , pr ola ctin,
ins ulin, ins ulin lik e gr owth f a ctor - I ( lGF- l) s om a tom edin- C
( SM- C) , ca lcitonin, leptin a nd the leptin der iv ed s hor t peptide
OB- 3 , m ela tonin, GLP- l or Gluca gon- lik e peptide- 1 a nd a na
logs ther eof , s uch a s ex ena tide, a lbiglutide, ta s poglutide, lir a
glutide a nd lix is ena tide, GiP, neur opeptide pituita r y a deny
la te cycla s e, GM- l ga nglios ide, ner v e gr owth f a ctor ( N GF) ,
na f a r elin, D - tr yp6) - LH RH , FGF, VEGF a nta gonis ts , leupr o
lide, inter f er on ( e. g. , 0t, [ 3 , y) low m olecula r weight hepa r in,
PY Y , LH RH a nta gonis ts , K er a tinocyte Gr owth Fa ctor
( K GF) , Glia l- D er iv ed N eur otr ophic Fa ctor ( GD N F) , ghr elin,
a nd ghr elin a nta gonis ts . Fur ther , in s om e a s pects , the peptide
or pr otein is s elected f r om a gr owth f a ctor , inter leuk in,
polypeptide v a ccine, enz ym e, endor phin, glycopr otein, lipo
pr otein, or a polypeptide inv olv ed in the blood coa gula tion
ca s ca de.
[ 0091] Cer ta in s hor t peptides com pos ed of a ppr ox im a tely
8 to 10 D - a m ino a cids des igna ted Allos ter a m er s pr oduced
by Allo s ter a Pha r m a lnc. , Q uebec, Ca na da , ha v e been s hown
to ha v e a n incr ea s ed degr ee of or a l bioa v a ila bility a s well a s
ex tended length of tim e in the blood s tr ea m . Such D - a m ino
a cid- conta ining peptides a r e pa r ticula r ly well s uited f or us e
with the pr es ent inv ention. Cycliz a tion, a s in cyclic PTH 1- 3 1
( N em eth 2008 ) , pr ov ides a nother wa y to r educe ga s tr ointes
tina l hydr olys is . Thus , in v a r ious a s pects , s hor t peptides con
ta ining non- na tur a lly occur r ing s tr uctur a l m odi? ca tions or
a m ino a cids a r e bes t s uited to the pr es ent inv ention. Peptides
com pr is ing les s tha n a bout 60, 50, 40, 3 0, 20, 15 or 10 a m ino
a cids a r e contem pla ted.
You might also like
- FACILE CROSSLINKINGDocument40 pagesFACILE CROSSLINKINGV Phanindra BoguNo ratings yet
- US20130131400 Etil AsetatDocument43 pagesUS20130131400 Etil AsetatDebby Theresia SilabanNo ratings yet
- Us 20140060005Document44 pagesUs 20140060005Victor Von DoomNo ratings yet
- US PatentDocument13 pagesUS Patenteni rohaniNo ratings yet
- Us 20130079618Document30 pagesUs 20130079618Fercalo AndreiNo ratings yet
- TM 10-8400-201-23&p General Repair Procedures For Clothing Oct. 2014Document612 pagesTM 10-8400-201-23&p General Repair Procedures For Clothing Oct. 2014GLOCK35100% (1)
- United States Patent (10) Patent N0.: US 8,091,836 B2Document8 pagesUnited States Patent (10) Patent N0.: US 8,091,836 B2werwer44345No ratings yet
- PNet Doc Status With ET Remarks 23 Dec 2011Document20 pagesPNet Doc Status With ET Remarks 23 Dec 2011niginpNo ratings yet
- Ribbon Bonding Tools and MethodsDocument29 pagesRibbon Bonding Tools and MethodstnchsgNo ratings yet
- Patent - Paul BabcockDocument25 pagesPatent - Paul BabcockHappyJoeNo ratings yet
- Us 20130218588Document46 pagesUs 20130218588david19775891No ratings yet
- Ray The On Pressure ShieldDocument26 pagesRay The On Pressure Shield...tho the name has changed..the pix remains the same.....No ratings yet
- Umted States Patent (10) Patent N0.2 US 8,415,942 B2: Fletcher (45) Date of Patent: Apr. 9, 2013Document19 pagesUmted States Patent (10) Patent N0.2 US 8,415,942 B2: Fletcher (45) Date of Patent: Apr. 9, 2013bonesisaliveNo ratings yet
- US20140182540Document47 pagesUS20140182540Amit PandeyNo ratings yet
- Dive Manual Rev 6 With CHG ADocument1,008 pagesDive Manual Rev 6 With CHG AJosé Cid MoragaNo ratings yet
- Us 20110139628Document9 pagesUs 20110139628Rahma Ama NuryantyNo ratings yet
- (12 Ulllted States Patent (10) Patent N0.: US 6,176,184 B1Document13 pages(12 Ulllted States Patent (10) Patent N0.: US 6,176,184 B1danceNo ratings yet
- Model 800 Viscometer Instruction ManualDocument23 pagesModel 800 Viscometer Instruction ManualNolanJohnsonNo ratings yet
- Unlted States Patent (10) Patent No.: US 8,314,576 B2Document42 pagesUnlted States Patent (10) Patent No.: US 8,314,576 B2avalosheNo ratings yet
- FGW P220HE2 Generator SpecificationsDocument1 pageFGW P220HE2 Generator SpecificationszavirkNo ratings yet
- Emerson-Valve Sizing Calculations (Traditional Method)Document21 pagesEmerson-Valve Sizing Calculations (Traditional Method)iqtorresNo ratings yet
- United States Patent (10) Patent No.: US 8,014,526 B2Document12 pagesUnited States Patent (10) Patent No.: US 8,014,526 B2sat258No ratings yet
- Stress Analysis Report AB118Document9 pagesStress Analysis Report AB118Pinak Projects100% (2)
- US20140178722A1Document21 pagesUS20140178722A1Afida MaelasariNo ratings yet
- Static Calculation For FansDocument4 pagesStatic Calculation For FansBadrul HishamNo ratings yet
- Automatic pricing of remotely hosted applicationsDocument26 pagesAutomatic pricing of remotely hosted applicationspankajpandeylkoNo ratings yet
- Us 20040075824Document18 pagesUs 20040075824presuraNo ratings yet
- Pukor 11-049e Priced Gsec Pdvsa Gas-Mega-Project 11058a-In-12 (P)Document43 pagesPukor 11-049e Priced Gsec Pdvsa Gas-Mega-Project 11058a-In-12 (P)avaisharmaNo ratings yet
- Viscosimetro OfiteDocument20 pagesViscosimetro OfitejomarlucaNo ratings yet
- Gel Strength (lbfl100 FT') : (12) United States Patent (10) Patent N0.2 US 6,955,220 B2Document9 pagesGel Strength (lbfl100 FT') : (12) United States Patent (10) Patent N0.2 US 6,955,220 B2b4rfNo ratings yet
- Financial Results & Limited Review For June 30, 2014 (Standalone) (Result)Document3 pagesFinancial Results & Limited Review For June 30, 2014 (Standalone) (Result)Shyam SunderNo ratings yet
- Max pipe span table for carbon steel, stainless steel & individual supportsDocument5 pagesMax pipe span table for carbon steel, stainless steel & individual supportsSeungmin PaekNo ratings yet
- Patent Application Publication (10) Pub. No.: US 2016/0348015 A1Document19 pagesPatent Application Publication (10) Pub. No.: US 2016/0348015 A1SebaNo ratings yet
- An Experimental and Theoretical Study of Planing Surfaces With Trim FlapsDocument63 pagesAn Experimental and Theoretical Study of Planing Surfaces With Trim Flapsمرتضی عزیزیNo ratings yet
- Oxygen Mask Regulator InspectionDocument16 pagesOxygen Mask Regulator Inspectionlutfi basithNo ratings yet
- Characterization of Casting Defect in Typical Castings of A Directionally Solidified SuperalloyDocument134 pagesCharacterization of Casting Defect in Typical Castings of A Directionally Solidified SuperalloyChun-Yi LinNo ratings yet
- TM 10-8400-203-23& P Indiv. Equip. Repair May 2010Document289 pagesTM 10-8400-203-23& P Indiv. Equip. Repair May 2010GLOCK35100% (1)
- Kheraj Pricelist 01.04.2012Document8 pagesKheraj Pricelist 01.04.2012anupam789No ratings yet
- Holden VL Series Service Manual SupplementDocument10 pagesHolden VL Series Service Manual SupplementmattNo ratings yet
- Ul Aire GD MideaDocument15 pagesUl Aire GD Mideaeduin gonzalezNo ratings yet
- CsLiLn Halide Scintillator Composition and Detection MethodsDocument17 pagesCsLiLn Halide Scintillator Composition and Detection MethodsBill HigginsNo ratings yet
- Research 2Document45 pagesResearch 2anaqiaisyahNo ratings yet
- Mixed Flow Voluta Case PumpsDocument12 pagesMixed Flow Voluta Case Pumpscesardiaz_ruizNo ratings yet
- United States PatentDocument17 pagesUnited States Patentbrar001No ratings yet
- US8578788Document30 pagesUS8578788chissyraezaNo ratings yet
- C Catg.t01Document113 pagesC Catg.t01akigujarNo ratings yet
- L1 - GSM Alcatel IntroductionDocument104 pagesL1 - GSM Alcatel Introductionsoyuz90No ratings yet
- Ulllted States Patent (10) Patent N0.: US 7,766,041 B2Document34 pagesUlllted States Patent (10) Patent N0.: US 7,766,041 B2scibduser001No ratings yet
- Solution: Conservation of EnergyDocument8 pagesSolution: Conservation of Energydist2235No ratings yet
- Adaptive Systems in Control and Signal Processing 1992: Selected Papers from the 4th IFAC Symposium Grenoble, France, 1 - 3 July 1992From EverandAdaptive Systems in Control and Signal Processing 1992: Selected Papers from the 4th IFAC Symposium Grenoble, France, 1 - 3 July 1992No ratings yet
- Wind Power in America's Future: 20% Wind Energy by 2030From EverandWind Power in America's Future: 20% Wind Energy by 2030No ratings yet
- Southern Marine Engineering Desk Reference: Second Edition Volume IFrom EverandSouthern Marine Engineering Desk Reference: Second Edition Volume INo ratings yet
- Endovascular Abdominal Aortic Repair- Endoleak Treatment: A Case-based ApproachFrom EverandEndovascular Abdominal Aortic Repair- Endoleak Treatment: A Case-based ApproachNo ratings yet
- Introduction to Linear Regression AnalysisFrom EverandIntroduction to Linear Regression AnalysisRating: 2.5 out of 5 stars2.5/5 (4)
- Current Therapy in EndodonticsFrom EverandCurrent Therapy in EndodonticsPriyanka JainNo ratings yet
- Code of Safe Working Practices for Merchant Seafarers Consolidated 2015 edition, including amendments 1-7From EverandCode of Safe Working Practices for Merchant Seafarers Consolidated 2015 edition, including amendments 1-7No ratings yet
- Taking The Mystery Out of The Maximum Allowable Carryover (MAC) Calculations For Cleaning ValidationDocument2 pagesTaking The Mystery Out of The Maximum Allowable Carryover (MAC) Calculations For Cleaning Validationjljimenez1969100% (3)
- Abuse Deterrent MarketDocument6 pagesAbuse Deterrent Marketmaneshdixit4312No ratings yet
- Brochure - LPI (Liberty)Document2 pagesBrochure - LPI (Liberty)maneshdixit4312No ratings yet
- Federal Cleanroom StandardsDocument6 pagesFederal Cleanroom StandardsmarkNo ratings yet
- AquaSolve As HandbookDocument16 pagesAquaSolve As Handbookmaneshdixit4312No ratings yet
- Animal Draft - GFI 234 - QBR For CMC Technical Section - 02242016 - Final - V2Document56 pagesAnimal Draft - GFI 234 - QBR For CMC Technical Section - 02242016 - Final - V2maneshdixit4312No ratings yet
- Critical Cleaning Guide: How To Select and Use Detergents For Critical Cleaning ApplicationsDocument20 pagesCritical Cleaning Guide: How To Select and Use Detergents For Critical Cleaning Applicationsmaneshdixit4312No ratings yet
- Critical Cleaning Guide: How To Select and Use Detergents For Critical Cleaning ApplicationsDocument20 pagesCritical Cleaning Guide: How To Select and Use Detergents For Critical Cleaning Applicationsmaneshdixit4312No ratings yet
- MAPP 5016.6 - Change in Hard Gelatin Capsule SupplierDocument2 pagesMAPP 5016.6 - Change in Hard Gelatin Capsule Suppliermaneshdixit4312No ratings yet
- RTR Guidance 2015 MayDocument25 pagesRTR Guidance 2015 Maymaneshdixit4312100% (1)
- Supac GuidelineDocument30 pagesSupac GuidelineSalman ShaikhNo ratings yet
- Zeta Potential - An Introduction in 30 MinutesDocument6 pagesZeta Potential - An Introduction in 30 MinutesFA AyNo ratings yet
- Enhancing ANDA Submissions: Working Toward First Cycle ApprovalDocument23 pagesEnhancing ANDA Submissions: Working Toward First Cycle Approvalmaneshdixit4312No ratings yet
- ANDA Litigation BasicsDocument27 pagesANDA Litigation Basicsmaneshdixit4312No ratings yet
- Estab Conditions Reporting by US FDADocument17 pagesEstab Conditions Reporting by US FDAmaneshdixit4312No ratings yet
- Impurity Guidance Table FDADocument4 pagesImpurity Guidance Table FDAmaneshdixit4312No ratings yet
- European Biosimilars Handbook enDocument54 pagesEuropean Biosimilars Handbook enmaneshdixit4312No ratings yet
- Alconox Applications in Pharmaceutical ManufacturingDocument12 pagesAlconox Applications in Pharmaceutical Manufacturingmaneshdixit4312No ratings yet
- FDA 2012 Biosimilar Quality ConsiderationDocument20 pagesFDA 2012 Biosimilar Quality Considerationmaneshdixit4312No ratings yet
- Stability Q ADocument16 pagesStability Q Amaneshdixit4312No ratings yet
- FDA 2013 Radhika Rajagopalan 1Document27 pagesFDA 2013 Radhika Rajagopalan 1maneshdixit4312No ratings yet
- Polyox Form Peo MatricesDocument7 pagesPolyox Form Peo Matricesmaneshdixit4312No ratings yet
- Magnesium Sterate Mixing in V BlenderDocument12 pagesMagnesium Sterate Mixing in V Blendermaneshdixit4312No ratings yet
- ANDA Filing Checklist for Completeness and AcceptabilityDocument12 pagesANDA Filing Checklist for Completeness and Acceptabilitymaneshdixit4312No ratings yet
- Guidance For IndustryDocument7 pagesGuidance For IndustryYash PatelNo ratings yet
- QBD ACE Case HistoryDocument92 pagesQBD ACE Case Historymaneshdixit4312No ratings yet
- Anatel PDFDocument4 pagesAnatel PDFmaneshdixit4312No ratings yet
- The M.T Taco Food Truck Business Plan Project Business Plan DraftDocument23 pagesThe M.T Taco Food Truck Business Plan Project Business Plan DraftAbhishek Prasad RoyNo ratings yet
- ParikalpDocument43 pagesParikalpManish JaiswalNo ratings yet
- BR A Consumables Catalog ElecDocument31 pagesBR A Consumables Catalog Elecdweil1552No ratings yet
- Development of The FaceDocument76 pagesDevelopment of The Facedr parveen bathla100% (1)
- Eb4069135 F enDocument13 pagesEb4069135 F enkalvino314No ratings yet
- Kathrein 80010375Document2 pagesKathrein 80010375klamar5No ratings yet
- Energy Analysis of Closed Systems BreakdownDocument31 pagesEnergy Analysis of Closed Systems Breakdownايات امجد امجدNo ratings yet
- Electronics Meet Animal BrainsDocument44 pagesElectronics Meet Animal BrainssherrysherryNo ratings yet
- 5 - Flight Recorder - OcrDocument19 pages5 - Flight Recorder - OcrtmhoangvnaNo ratings yet
- ElectrochemistryDocument24 pagesElectrochemistryZainul AbedeenNo ratings yet
- Steel 17-4PH MmpdsDocument18 pagesSteel 17-4PH MmpdsManoj ManoharanNo ratings yet
- MSDS Corn CobDocument2 pagesMSDS Corn CobMUHAMMAD YOGA BRILLIANTONo ratings yet
- Hart Transmitter Calibration: Application NoteDocument8 pagesHart Transmitter Calibration: Application NoteThulasi Raman KowsiganNo ratings yet
- Nsf-Ansi 55 PDFDocument56 pagesNsf-Ansi 55 PDFJawwad AhmedNo ratings yet
- AWWA M28 Rehabilitation of Water Mains 3rd Ed 2014Document133 pagesAWWA M28 Rehabilitation of Water Mains 3rd Ed 2014millini67% (3)
- 2017 Scoring Materials Math g6Document119 pages2017 Scoring Materials Math g6KRANo ratings yet
- Masterbrand Guidelines - September 2012: Confidential - For Internal Use OnlyDocument35 pagesMasterbrand Guidelines - September 2012: Confidential - For Internal Use OnlyDemerson CamposNo ratings yet
- Discrete Variable Probability Distribution FunctionsDocument47 pagesDiscrete Variable Probability Distribution FunctionsJanine CayabyabNo ratings yet
- 692pu 6 6Document1 page692pu 6 6Diego GodoyNo ratings yet
- 32776Document6 pages32776Muqtar KhanNo ratings yet
- Plug & Socket TypesDocument49 pagesPlug & Socket TypeskerissaktiemasNo ratings yet
- Advanced Technologies of CDQ Plant Advanced Technologies of CDQ PlantDocument12 pagesAdvanced Technologies of CDQ Plant Advanced Technologies of CDQ Plant조기현No ratings yet
- Contemporary Philippine Arts From The Regions: Quarter 3Document15 pagesContemporary Philippine Arts From The Regions: Quarter 3Ackie Inacay RosarioNo ratings yet
- Poisoning: Selenium in LivestockDocument4 pagesPoisoning: Selenium in Livestockdianarbk otuNo ratings yet
- Nutrition and Athletic PerformanceDocument16 pagesNutrition and Athletic PerformancemarisolNo ratings yet
- Operating Systems: Chapter 3: DeadlocksDocument46 pagesOperating Systems: Chapter 3: DeadlocksManoel Katlib100% (1)
- Scheme of Valuation and Key for Transportation Engineering ExamDocument3 pagesScheme of Valuation and Key for Transportation Engineering ExamSivakumarNo ratings yet
- Railway Electrification Projects Budget 2019-20Document9 pagesRailway Electrification Projects Budget 2019-20Muhammad Meraj AlamNo ratings yet
- Toaz - Info Chemtech Reviewer PRDocument1 pageToaz - Info Chemtech Reviewer PRchristy janioNo ratings yet
- Wire Rope: - Bright - 6 X 19 - Fibre CoreDocument8 pagesWire Rope: - Bright - 6 X 19 - Fibre CoreQuynh NguyenNo ratings yet