Professional Documents
Culture Documents
ScP019 Chemical Equations 1
Uploaded by
ORBeducationCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
ScP019 Chemical Equations 1
Uploaded by
ORBeducationCopyright:
Available Formats
Balancing Equations
Writing balanced symbol equations
There are four stages to writing a full equation for a reaction:
1. Write out the word equation
2. Work out the formulae for all elements and compounds present
3. Balance the equation
4. Add information about the state of each chemical (solid, liquid, gas or aqueous solution).
This worksheet is concerned with the third task – balancing symbol equations.
Example 1 - The reaction between magnesium and oxygen
Magnesium + Oxygen Magnesium Oxide
We can work out (using valency or otherwise) that the formula for magnesium oxide is MgO.
We need to remember that oxygen is a diatomic molecule and hence has the formula O2.
We can now begin our symbol equation:
Magnesium + Oxygen Magnesium Oxide
Mg + O2 Mg0
The next stage is to look at the number of atoms of each type of element on either side of the equation. If we start with one
magnesium atom, we must finish the reaction with one. If we start with two oxygen atoms, we must also end up with two.
Mg + O2 Mg0
Mg O Mg
+
O O
We can see from the diagram that there is the same number of magnesium atoms on either side of the arrow, but the oxygen
atoms are not balanced. We cannot introduce a single oxygen atom to the right hand side. We can only introduce a whole
magnesium oxide group. We do this by placing a 2 before the MgO formula.
Mg + O2 2Mg0
Mg + O Mg
Mg
O O
O
We now have two oxygen atoms on each side of the equation, but the magnesium atoms no longer match. We have to introduce
one more magnesium atom to the left hand side, We do this by placing a 2 in front of the magnesium symbol.
Magnesium + Oxygen Magnesium Oxide
2Mg + O2 2Mg0
Mg Mg
Mg + O Mg
O
O O
Our symbol equation is now balanced.
ORB Education Quality Teaching Resources
ORB Education Visit http://www.orbedu.com for the full, editable versions with solutions. ScP019 - Chemical Equations 1
Task 1
Use the diagrams to help balance these symbol equations:
a. Sodium + Chlorine Magnesium Oxide
Cl Na
Na Na Na
+ Cl Cl
Cl
Balance
Na + Cl2 NaCl
this
b. Aluminum + Oxygen Aluminum Oxide
Al O
O O
Al
Al O
O O
Al + Al
O
O O Al
O O
O
Balance
Al + O2 Al2O3
this
Task 2
Use diagrams or any other method to help balance these symbol equations:
a. Ca + O2 Ca0
b. Li + F2 LiF
c. Mg + Br2 MgBr2
d. K + O2 K20
e. Al + Cl2 AlCl3
f. Fe + O2 Fe203
g. H2 + O2 H2O
h. H2 + Cl2 HCl
i. S + O2 SO2
j. C + H2 CH4
k. Ag + O2 Ag2O
l. Ca + HCl CaCl2 + H2
m. Mg + HCl MgCl2 + H2
n. Na + HCl NaCl + H2
o. Al + HCl AlCl3 + H2
ORB Education Quality Teaching Resources
ORB Education Visit http://www.orbedu.com for the full, editable versions with solutions. ScP019 - Chemical Equations 1
You might also like
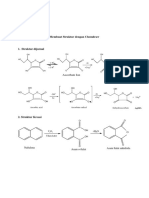
- Crystals: Incidental Polymorphism, Non-Isomorphic and Isomorphic Substitution in Calcium-Valine Coordination PolymersDocument12 pagesCrystals: Incidental Polymorphism, Non-Isomorphic and Isomorphic Substitution in Calcium-Valine Coordination Polymersmike7138No ratings yet
- Customer HYDRO-FOIL S8 SystemDocument21 pagesCustomer HYDRO-FOIL S8 SystemLong Nhật PhanNo ratings yet
- Kami Export - Natalie Hurst - Chemical Equations Worksheet QuizDocument1 pageKami Export - Natalie Hurst - Chemical Equations Worksheet QuizNatalie HurstNo ratings yet
- Kami Export - Natalie Hurst - Chemical Equations Worksheet QuizDocument1 pageKami Export - Natalie Hurst - Chemical Equations Worksheet QuizNatalie HurstNo ratings yet
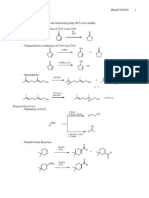
- A Useful Grignard Reaction: The Synthesis of Dyes: Chemistry 223LDocument2 pagesA Useful Grignard Reaction: The Synthesis of Dyes: Chemistry 223LRamona AndronesiNo ratings yet
- 3 MOD StudentsDocument20 pages3 MOD StudentsMattNo ratings yet
- SelectivityDocument4 pagesSelectivitySamik BiswasNo ratings yet
- Advanced Organic Reactions 2000 - WarrenDocument174 pagesAdvanced Organic Reactions 2000 - Warrenshiv57100% (3)
- Membuat Struktur Dengan Chemdraw: Nama: Riza Gustina NPM: A1F015009Document1 pageMembuat Struktur Dengan Chemdraw: Nama: Riza Gustina NPM: A1F015009isnaini safitriNo ratings yet
- Online Edition - Digital AccessDocument1 pageOnline Edition - Digital Access7dwg75s8mgNo ratings yet
- 03 - Chemical Bonding and Structure CORNELLDocument21 pages03 - Chemical Bonding and Structure CORNELLGeorge SolomouNo ratings yet
- Alcohol and Phenol - 18290367 - 2023 - 06 - 20 - 12 - 13Document6 pagesAlcohol and Phenol - 18290367 - 2023 - 06 - 20 - 12 - 13telate6613No ratings yet
- (+) - Epoxyquinols A and B (TKGP-100417) M. Shoji, Y. Hayashi: ActivityDocument2 pages(+) - Epoxyquinols A and B (TKGP-100417) M. Shoji, Y. Hayashi: ActivityPercival GalahadNo ratings yet
- R É ArrangementDocument68 pagesR É ArrangementFakher NtichaNo ratings yet
- 3 - Hardness - Applied ChemistryDocument15 pages3 - Hardness - Applied ChemistryHarshit RajNo ratings yet
- BFD - BFDDocument3 pagesBFD - BFDSendra Naq AyodanceNo ratings yet
- Experiment 5: Preparation of An, Unsaturated Ketone Via Michael Addition and Aldol Condensation ReactionsDocument10 pagesExperiment 5: Preparation of An, Unsaturated Ketone Via Michael Addition and Aldol Condensation ReactionsNur AthirahNo ratings yet
- Slag LectureDocument39 pagesSlag LectureJojo HanyNo ratings yet
- 24 Nov 06Document4 pages24 Nov 06Subham NandiNo ratings yet
- Acidity and Basicity of Zeolites - A Fundamental ApproachDocument49 pagesAcidity and Basicity of Zeolites - A Fundamental ApproachHenrique SouzaNo ratings yet
- Year 10 Balancing Equations - Level 2 Year 10 Balancing Equations - Level 2Document2 pagesYear 10 Balancing Equations - Level 2 Year 10 Balancing Equations - Level 2Gaming TriadNo ratings yet
- Chp-11-Electrochemistry, Redox Reaction (E)Document11 pagesChp-11-Electrochemistry, Redox Reaction (E)Khe ManshuNo ratings yet
- Reactions of 2-Bromo-4-Phenylcyclohexanols With Base and Silver OxideDocument13 pagesReactions of 2-Bromo-4-Phenylcyclohexanols With Base and Silver OxideAmar dattaNo ratings yet
- miscon spot bond (1) - 已导出Document2 pagesmiscon spot bond (1) - 已导出Li JefferyNo ratings yet
- IMO55 2021 T1 Solutions EngDocument11 pagesIMO55 2021 T1 Solutions EngLazar SavicNo ratings yet
- Answers To Science Focus 3 Coursebook QuestionsDocument61 pagesAnswers To Science Focus 3 Coursebook QuestionsrobouNo ratings yet
- Atomic Structure and The Periodic Table Foundation Revision Activity MatDocument4 pagesAtomic Structure and The Periodic Table Foundation Revision Activity MatHồng Ngọc VõNo ratings yet
- Problems 4 ADocument1 pageProblems 4 ALngNo ratings yet
- Activity Series & BR I N CL H O FDocument2 pagesActivity Series & BR I N CL H O FYob BeeNo ratings yet
- Chemistry 6941, Fall 2007 Synthesis Problems I - Key Dr. Peter NorrisDocument9 pagesChemistry 6941, Fall 2007 Synthesis Problems I - Key Dr. Peter NorrisQuốc NguyễnNo ratings yet
- 2D C11) Type of ReactionDocument1 page2D C11) Type of Reactionlaurencrowe08No ratings yet
- Nanh (1 Equiv.) 2. CH - CH - I H, Lindlar Catalyst Mcpba CH Li Cubr TSCL Pyridine NacnDocument12 pagesNanh (1 Equiv.) 2. CH - CH - I H, Lindlar Catalyst Mcpba CH Li Cubr TSCL Pyridine NacnpNo ratings yet
- Module-3 (Part-I)Document218 pagesModule-3 (Part-I)Prajay GNo ratings yet
- Chemical Reaction and Balancing Chemical EquationDocument36 pagesChemical Reaction and Balancing Chemical EquationChelsia Venice MorilloNo ratings yet
- Selective Determination of Fe (III) in Fe (II) Samples by UV-spectrophotometry With The Aid of Quercetin and MorinDocument8 pagesSelective Determination of Fe (III) in Fe (II) Samples by UV-spectrophotometry With The Aid of Quercetin and MorinSirisha NNo ratings yet
- Organometallic ReactionsDocument7 pagesOrganometallic Reactionszahara99121No ratings yet
- Year 10 Balancing Equations - Level 1 Year 10 Balancing Equations - Level 1Document2 pagesYear 10 Balancing Equations - Level 1 Year 10 Balancing Equations - Level 1Gaming TriadNo ratings yet
- Composition AxesDocument3 pagesComposition AxesEDGARDO RAUL ROQUE CHOQUENo ratings yet
- Carbonyl Chemistry Tutorial #9 2018-2019 AnswersDocument9 pagesCarbonyl Chemistry Tutorial #9 2018-2019 AnswersZoe NorvilleNo ratings yet
- Solution Felder 9.16Document3 pagesSolution Felder 9.16Alya Sholikhatul100% (1)
- Lipase MechanismDocument2 pagesLipase Mechanismrendy jwjNo ratings yet
- Chem 16 LabDocument19 pagesChem 16 LabDiyanikaNo ratings yet
- We Are Intechopen, The World'S Leading Publisher of Open Access Books Built by Scientists, For ScientistsDocument17 pagesWe Are Intechopen, The World'S Leading Publisher of Open Access Books Built by Scientists, For ScientistsWissal ZitouniNo ratings yet
- Ions: How Are They Formed?Document23 pagesIons: How Are They Formed?Charlotte Rovelyn LorenzoNo ratings yet
- Igcse ChemistryDocument2 pagesIgcse Chemistrysiddhi thakkarNo ratings yet
- Chemical Bonding Practice Chemical Bonding PracticeDocument2 pagesChemical Bonding Practice Chemical Bonding PracticeBHAWANA SINGHNo ratings yet
- Green Chemistry PointsDocument45 pagesGreen Chemistry PointsPabloNo ratings yet
- Redox - Super Live Session: Contact Number: 9667591930 / 8527521718Document2 pagesRedox - Super Live Session: Contact Number: 9667591930 / 8527521718Tushar SahaNo ratings yet
- REACTIVO 2 TermodinamicaDocument5 pagesREACTIVO 2 TermodinamicaCalvin JacobNo ratings yet
- Chemical Nomenclature Stations AP ChemistryDocument8 pagesChemical Nomenclature Stations AP ChemistryJohnny MoyaNo ratings yet
- Analysis of Cima PaintingOptDocument11 pagesAnalysis of Cima PaintingOptmaryjaneapuadaNo ratings yet
- Problem Set 7 - Key: Due Tuesday, November 15, 2016Document9 pagesProblem Set 7 - Key: Due Tuesday, November 15, 2016Trần Nguyễn Quỳnh NhưNo ratings yet
- ScP033 Mixed Power PointsDocument24 pagesScP033 Mixed Power PointsORBeducationNo ratings yet
- ScP046 Graphing ToolboxDocument2 pagesScP046 Graphing ToolboxORBeducationNo ratings yet
- ScP036 Chem Lab ToolboxDocument1 pageScP036 Chem Lab ToolboxORBeducationNo ratings yet
- ScP037 Circuits ToolboxDocument2 pagesScP037 Circuits ToolboxORBeducationNo ratings yet
- ScP034 Atoms Power PointsDocument9 pagesScP034 Atoms Power PointsORBeducationNo ratings yet
- ScP045 Scales ToolboxDocument2 pagesScP045 Scales ToolboxORBeducationNo ratings yet
- ScP043 ReproductionDocument1 pageScP043 ReproductionORBeducationNo ratings yet
- ScP051 CellsDocument1 pageScP051 CellsORBeducationNo ratings yet
- 6th Central Pay Commission Salary CalculatorDocument15 pages6th Central Pay Commission Salary Calculatorrakhonde100% (436)
- ScP049 Practical Task Sheets 1Document2 pagesScP049 Practical Task Sheets 1ORBeducationNo ratings yet
- ScP050 Discovering Solar SystemDocument1 pageScP050 Discovering Solar SystemORBeducationNo ratings yet
- ScP024 Cells ResearchDocument1 pageScP024 Cells ResearchORBeducationNo ratings yet
- ScP052 Vocab ResourcesDocument1 pageScP052 Vocab ResourcesORBeducationNo ratings yet
- ScP044 Human SystemsDocument2 pagesScP044 Human SystemsORBeducation0% (1)
- ScP027 Animals ResearchDocument1 pageScP027 Animals ResearchORBeducationNo ratings yet
- ScP039 Practical Alternatives 1Document1 pageScP039 Practical Alternatives 1ORBeducationNo ratings yet
- ScP042 Ecology ActivitiesDocument2 pagesScP042 Ecology ActivitiesORBeducationNo ratings yet
- ScP029 PlanetsDocument1 pageScP029 PlanetsORBeducationNo ratings yet
- ScP030 Energy ResourcesDocument1 pageScP030 Energy ResourcesORBeducationNo ratings yet
- ScP031 States LADocument2 pagesScP031 States LAORBeducation100% (4)
- ScP026 Systems ResearchDocument1 pageScP026 Systems ResearchORBeducationNo ratings yet
- ScP025 Organs ResearchDocument1 pageScP025 Organs ResearchORBeducationNo ratings yet
- ScP028 Biology JigsawsDocument1 pageScP028 Biology JigsawsORBeducationNo ratings yet
- ScP032 Elements LADocument1 pageScP032 Elements LAORBeducationNo ratings yet
- ScP023 ForcesDocument1 pageScP023 ForcesORBeducationNo ratings yet
- ScP022 Light RaysDocument1 pageScP022 Light RaysORBeducationNo ratings yet
- ScP021 ColorDocument2 pagesScP021 ColorORBeducationNo ratings yet
- ScP018 BiologyDocument1 pageScP018 BiologyORBeducationNo ratings yet
- ScP020 Chemical Equations 2Document1 pageScP020 Chemical Equations 2ORBeducationNo ratings yet
- Unit 1 Organic Chemistry: 1. Name The Following MoleculeDocument25 pagesUnit 1 Organic Chemistry: 1. Name The Following MoleculeGabby PortilloNo ratings yet
- Olympiad 2007 r1 Mark SchemeDocument7 pagesOlympiad 2007 r1 Mark SchemeJackieWilsonNo ratings yet
- (VCE Chemistry) 2013 LisaChem Unit 1 Exam and SolutionsDocument36 pages(VCE Chemistry) 2013 LisaChem Unit 1 Exam and SolutionslogophileNo ratings yet
- Enhanced Hybrid Module S7 Q1 M2 Week 2 and 3.editedDocument15 pagesEnhanced Hybrid Module S7 Q1 M2 Week 2 and 3.editedHannah Joy LontayaoNo ratings yet
- Home School FORM 3 CHEMISTRYDocument37 pagesHome School FORM 3 CHEMISTRYgabrielsuva6No ratings yet
- Lecture 8 - Reading and Writing in ChemistryDocument14 pagesLecture 8 - Reading and Writing in ChemistryEwartNo ratings yet
- Glinka Problems in General Chemistry MirDocument290 pagesGlinka Problems in General Chemistry MirMaurizio D'IppolitoNo ratings yet
- Naming Compounds LessonDocument2 pagesNaming Compounds LessonDarlene VoogtNo ratings yet
- Ionic BondingDocument51 pagesIonic BondingAaditya MKNo ratings yet
- STPM Trials 2009 Chem P2 (N Sembilan), Pra-2010Document11 pagesSTPM Trials 2009 Chem P2 (N Sembilan), Pra-2010Looi Chui YeanNo ratings yet
- Quarter 1 AssessmentDocument6 pagesQuarter 1 AssessmentlkNo ratings yet
- Index of H DeficiencyDocument2 pagesIndex of H DeficiencyVarun Singh KushwahNo ratings yet
- 2020-OrgMol-Intro To Organic Chemistry - Part 1Document11 pages2020-OrgMol-Intro To Organic Chemistry - Part 1azraelNo ratings yet
- Science7 Q1 Mod2E Compounds Version3Document24 pagesScience7 Q1 Mod2E Compounds Version3Bryzel Cortes100% (2)
- 0620 w08 QP 03Document25 pages0620 w08 QP 03Hubbak KhanNo ratings yet
- PreviewpdfDocument30 pagesPreviewpdfPatofnaPatofnicNo ratings yet
- S5 ActivityDocument3 pagesS5 ActivityMaku MichaelNo ratings yet
- Unit 14 Structure Elucidation by Integrated Spectroscopic MethodsDocument25 pagesUnit 14 Structure Elucidation by Integrated Spectroscopic Methodscbnnoon222No ratings yet
- IGCSE Chemistry CalculationsDocument27 pagesIGCSE Chemistry CalculationsMartin MulengaNo ratings yet
- Chemical Basis of LifeDocument1 pageChemical Basis of LifeChris_Barber09100% (2)
- Hrvoj Vančik (Auth.) - Basic Organic Chemistry For The Life Sciences-Springer International Publishing (2014) PDFDocument188 pagesHrvoj Vančik (Auth.) - Basic Organic Chemistry For The Life Sciences-Springer International Publishing (2014) PDFPako GomezNo ratings yet
- 5070 Learner Guide (For Examination From 2023)Document43 pages5070 Learner Guide (For Examination From 2023)Zainab Hassan0% (2)
- Grade 9 Science Chemistry 4 DLPDocument9 pagesGrade 9 Science Chemistry 4 DLPManongdo AllanNo ratings yet
- Test 3Document6 pagesTest 3Davies MasumbaNo ratings yet
- Atoms and MoleculesDocument10 pagesAtoms and MoleculesUtsav Kumar MathurNo ratings yet
- General Chemistry - Acids and BaseDocument14 pagesGeneral Chemistry - Acids and BasePaul JacalanNo ratings yet
- Class 8th Chemistry Icse NotesDocument23 pagesClass 8th Chemistry Icse NotesSANDEEP SINGH0% (1)
- Chemical Equation Notes - TeacherDocument18 pagesChemical Equation Notes - TeachersmedificationNo ratings yet
- 11 Chemistry Notes Chapter 12Document16 pages11 Chemistry Notes Chapter 12avni AroraNo ratings yet
- Chemistry 101 Complete NotesDocument323 pagesChemistry 101 Complete NotesEnerolisa Paredes100% (1)