Professional Documents
Culture Documents
MEG Consent
Uploaded by
kafka730 ratings0% found this document useful (0 votes)
19 views11 pagesA consent form for my MEG study; could be useful for researchers new to the modality.
Copyright
© Attribution Non-Commercial (BY-NC)
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentA consent form for my MEG study; could be useful for researchers new to the modality.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
19 views11 pagesMEG Consent
Uploaded by
kafka73A consent form for my MEG study; could be useful for researchers new to the modality.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 11
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 1 of 11
Subject Identification
Protocol Title: Brain dynamics of oxytocin
Principal Investigator: Lilianne R. Mujica-Parodi (Strey), Ph.D.
Site Principal Investigator:
Description of Subject Population: Healthy Adults
CONSENT F ORM F OR ME G
About this consent form
Please read this form carefully. It tells you important information about a research study. A
member of our research team will also talk to you about taking part in this research study.
People who agree to take part in research studies are called 'subjects. This term will be used
throughout this consent form.
Partners HealthCare System is made up of Partners hospitals, health care providers, and
researchers. In the rest of this consent form, we refer to the Partners system simply as
'Partners.
If you have any questions about the research or about this form, please ask us. Taking part in
this research study is up to you. If you decide to take part in this research study, you must sign
this form to show that you want to take part. We will give you a signed copy of this form to
keep.
---------------------------------------------------------------------------------------------------------------------
A description of this clinical trial will be available on http://www.ClinicalTrials.gov, as required
by U.S. Law. This website will not include information that can identify you. At most, the
website will include a summary of the results. You can search this website at any time.
---------------------------------------------------------------------------------------------------------------------
Why is this research study being done?
The purpose of the study is to understand how oxytocin, a substance naturally produced by
everyone`s body, affects brain and behavior. To do this, we will do MEG
(magnetoencephalography; a type of scan that lets us map the activity of the brain by measuring
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 2 of 11
Subject Identification
the small magnetic fields that are produced by brain activity) scanning to look at how oxytocin
affects your brain activity.
Oxytocin has effects on social behavior, such as bonding between parents and their children or
between friends. To study behavioral effects, oxytocin is usually given to people as a nasal
spray under the label 'Syntocinon Nasal Spray. A Iorm oI oxytocin that is administered by IV
(intravenously; into a vein) is approved by the U.S. Food and Drug Administration (FDA) to
trigger labor in women during the end of their pregnancies. The FDA has not approved the nasal
spray form of oxytocin nor is oxytocin approved for affecting behavior.
This research study will compare Syntocinon Nasal Spray to placebo. The placebo looks exactly
like Syntocinon Nasal Spray, but contains no oxytocin. At some time during the study, we will
give you Syntocinon. At another time, we will give you placebo. Placebos are used in research
studies to see if the results are due to the study drug or due to other reasons.
This study has two parts: an MEG part and an MRI (magnetic resonance imaging; using a strong
magnet and radio waves to take detailed pictures of the body) part. This consent form is only for
the MEG part of the study. If you choose to take part in the MRI part of the study, you will sign a
separate consent form that describes what will be done during the MRI study.
We are asking you to take part in this study because you are a healthy volunteer.
About 50 people will take part in this research study, all at Massachusetts General Hospital
(MGH).
The Office of Naval Research is paying for the study to be done.
How long will I take part in this research study?
It will take you up to a week to complete this part of the study. During this time, we will ask you
to make up to two study visits, with the option to add an additional two study visits. The study
visits will not be more than 1 week apart. The visits for this study will take place at Building
149, at the Charlestown Navy Yard.
What will happen in this research study?
If you choose to take part in this study, we will ask you to read and sign this consent form before
we do any study procedures.
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 3 of 11
Subject Identification
We will assign you by chance (like a coin toss) to Syntocinon or placebo. If you receive
Syntocinon at the first visit, you will receive placebo at the second visit. If you receive placebo at
the first visit, you will receive Syntocinon at the second. Neither you nor the study doctor will be
able to choose whether you receive Syntocinon or placebo first. Both the Syntocinon Nasal
Spray and placebo will be reIerred to as study drug` Irom here on in the consent Iorm. You and
the study doctor won`t know which study group you are in, but the study doctor can Iind out iI
necessary.
Study Visit 1
This visit will take about 90 minutes.
Before the Scan
Before the scan, we will do some screening procedures. These tests and procedures are done to
see if you qualify to take part in this research study. The study doctor will review the results of
these tests and procedures. II you don`t qualiIy, the study doctor will tell you why. At this part
of the visit, we will:
Have you do a smell test so we can make sure your sense of smell works properly and
that your nose is not stuffed up. For example, you might be asked to tell the difference
between orange and vanilla odors.
Ask about how you are feeling and medical and emotional history.
Ask questions to make sure it is safe for you to have the MRI.
Test your urine for certain drugs, including illegal drugs such as cocaine and marijuana.
If your urine shows you have taken any oI these drugs, you can`t be in this study. The
results of the urine drug test will not become part of your medical record. These test
results will, however, remain part of your confidential study record.
Test your urine for pregnancy, if you are a female able to become pregnant. Pregnant
women cannot take part in this research study.
If you are still eligible to take part in this study, we will give you several puffs of the study drug
into your nose before the scan.
ME G Scan
The scan will take up to an hour. We will have you sit in a chair or lay on a bed. We will slide
head into the helmet-shaped bottom of the MEG. The MEG device has magnetic field sensors
that can detect the weak magnetic fields produced by your brain activity.
The MEG will not make any noise. Before we start the MEG testing, 4-5 small coils will be
taped to the skin of your head. Attaching the coils to your head will not hurt. Small electrical
currents will be passed through the coils once you are in the machine; this will allow us to
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 4 of 11
Subject Identification
position each coil on the right part oI your head so the sensors can pick up your brain`s activity.
You will not feel any of the currents.
The MEG is done in a special magnetically shielded room (MSR). Because the magnetic fields
produced by your brain activity are very small, they can be disrupted if there is any other
magnetic activity nearby. Because the MEG is always on, you will be asked to remove all metal
items before entering the MSR, as these objects may damage the sensors. For example, belts,
keys, watches, coins, hair barrettes and pins, eyeglasses, and pieces of clothing with magnetic
parts should be removed before you enter the MSR. Also, to prevent any magnetic dust from
getting into the MSR, you will remove shoes before entering. Objects such as cell phones,
pagers, cameras, flashlights, or any other electrical equipment are not allowed inside the MSR.
During the scan, we will use an eye-tracker, which uses a camera to record the movement of
your eyeswhile we gather data. During the scan, you will first rest for about 10 minutes. Then,
you will play a game where you will decide how to split a monetary reward between you and one
or more players. It is very important that you remain still and avoid moving your head during
recordings.
Between recordings, you will be able to speak by microphone with a member of the study staff
performing the scan.
Study Visit 2
This visit will be the same as visit 1 and will take about 90 minutes. You will repeat the
screening procedures and the MEG that were done in visit 1. At visit 2 you will receive
whichever study drug you did not receive at visit 1.
Storage and Use of your Study Information
We will label all your study information with a code instead of your name. The key to the code
connects your name to your study information. We will keep the key to the code here at Partners.
No one outside Partners will know which information is yours.
You may be given the option to undergo a second set of MEG scans in order to optimize image
quality. You will be given a full explanation of the study after you complete all study procedures.
What are the risks and possible discomforts from being in this research
study?
Risk of Syntocinon Nasal Spray
The side effects of Syntocinon Nasal Spray when used to induce labor are
Nausea and vomiting
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 5 of 11
Subject Identification
Bleeding
Abnormal heartbeats
Skin rashes
Fall in blood pressure (causing dizziness, lightheadedness, Ieeling Iaint)
DiIIiculty breathing
Shock (blood pressure too low to maintain blood supply to the tissues)
Rarely, adverse effects of oxytocin have been reported during labor, including nose bleeds,
tingling in the mouth or throat, lightheadedness, headache, nausea, and changes in heartbeat.
In studies more similar to this study, in which Syntocinon was used during behavioral testing or
during scans, the side effects are not the same as those reported in people using Syntocinon to
induce labor. For example, in 647 male subjects taking part in a similar study that used the same
dose of Syntocinon Nasal Spray, two subjects felt irritable. One subject reported stomach
cramps and one subject reported an increased sensitivity to smells. In most cases, you will not be
able to tell whether you are being given oxytocin or the placebo. While the side effects to
oxytocin are minimal and unlikely, a study doctor will be available at all times throughout the
research study in case you have any difficulties, questions, or concerns.
Risk of Allergic Reaction
As with any drug, an allergic reaction can occur. Allergic reactions can be mild or serious, and
can even result in death in some cases. Common symptoms of an allergic reaction are rash,
itching, skin problems, swelling of the face and throat, or trouble breathing. If you think you are
having an allergic reaction, tell the study doctor right away.
Risks/Discomforts of the ME G Scan
There are no known significant risks to having an MEG.
People who have electrically, magnetically, or mechanically activated implants (such as cardiac
pacemakers), or those with metal in their body (such as clips on blood vessels in their brain, steel
pins from root canals, or other metal fragments) cannot have an MEG because these devices will
disturb the MEG signal, and may cause damage to the MEG sensors. However, you may have an
MEG if you have metal dental fillings.
Risks to an Embryo or Fetus
Oxytocin can trigger labor in pregnant women. The effect of Syntocinon Nasal Spray on an
embryo or fetus (developing baby still in the womb), or on a breastfeeding infant, may be
harmful. Because of the risks, women cannot take part in this study if they are:
Pregnant
Trying to become pregnant
Breastfeeding
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 6 of 11
Subject Identification
If you are a menopausal woman and have not had a period for the past 12 months or more, you
will not need to have a pregnancy test. Also, if you have had any well-documented method of
surgical sterilization, you will not need to have a pregnancy test. Methods of surgical
sterilization include having had a hysterectomy (removal of the uterus with or without the
ovaries), bilateral oophorectomy (removal of both ovaries), a tubal ligation (having your tubes
tied), or transvaginal occlusion (plugging the opening of the tubes with a coil). All other female
subjects must have a negative pregnancy test before receiving the study drug.
Emotional Risks
There may be some temporary emotional discomfort involved in taking tests and answering
personal questions.
Other Risks
There may be other risks or side effects of Syntocinon Nasal Spray that are not yet known.
What are the possible benefits from being in this research study?
This study will not benefit you. Others may benefit in the future from what we learn in this
study. We hope to find new information that will help scientists to understand how a
person`s own, naturally-produced, oxytocin affects his or her behaviorsuch as bonding to a
child, partner, or friend.
&DQ,VWLOOJHWPHGLFDOFDUHZLWKLQ3DUWQHUVLI,GRQWWDNHSDUWLQWKLV
research study, or if I stop taking part?
Yes. Your decision won`t change the medical care you get within Partners now or in the Iuture.
There will be no penalty, and you won`t lose any beneIits you receive now or have a right to
receive.
Taking part in this research study is up to you. You can decide not to take part. If you decide to
take part now, you can change your mind and drop out later. We will tell you if we learn new
information that could make you change your mind about taking part in this research study.
What should I do if I want to stop taking part in the study?
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 7 of 11
Subject Identification
If you take part in this research study, and want to drop out, you should tell us. We will make
sure that you stop the study safely. We will also talk to you about follow-up care, if needed.
It is possible that we will have to ask you to drop out before you finish the study. If this
happens, we will tell you why. We will also help arrange other care for you, if needed.
Will I be paid to take part in this research study?
During this study, you will be playing a strategic gambling game in which you will have the
opportunity to win actual money, ranging from $40 to $100. If you do not complete the study, we
will pay you $10 for each hour you completed. You may be given the option to undergo a second
set of MEG scans in order to optimize image quality. If you choose to repeat the protocol, you
will have the opportunity to win an additional $40-$100.
Legally, you can be paid only if you are a US citizen, a legal resident alien (e.g. possess a green
card), or have a work eligible visa sponsored by the paying institution.
What will I have to pay for if I take part in this research study?
Study funds will pay for the study drug, the MEG recordings, and all other procedures at the
study visits.
Charges for any ongoing or routine medical care you receive outside this study will be billed to
you or to your insurance company in the usual way. You will be responsible for any deductibles
or co-payments required by your insurer for your routine medical care.
What happens if I am inj ured as a result of taking part in this research
study?
We will offer you the care needed to treat any injury that directly results from taking part in this
research study. We reserve the right to bill your insurance company or other third parties, if
appropriate, for the care you get for the injury. We will try to have these costs paid for, but you
may be responsible for some of them. For example, if the care is billed to your insurer, you will
be responsible for payment of any deductibles and co-payments required by your insurer.
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 8 of 11
Subject Identification
Injuries sometimes happen in research even when no one is at fault. There are no plans to pay
you or give you other compensation for an injury, should one occur. However, you are not
giving up any of your legal rights by signing this form.
If you think you have been injured or have experienced a medical problem as a result of taking
part in this research study, tell the person in charge of this study as soon as possible. The
researcher's name and phone number are listed in the next section of this consent form.
If I have questions or concerns about this research study, whom can I
call?
You can call us with your questions or concerns. Our telephone numbers are listed below. Ask
questions as often as you want.
Lilianne R. Mujica-Parodi, Ph.D. is the person in charge of this research study. You can call her
at 917-669-3934 M-F 9-5. The physicians are Dr. Karleyton Evans, who can be reached at 617-
724-0244 M-F 9-5, Dr. Bradford Dickerson, who can be reached at 617-726-5571 M-F 9-5, and
Dr. Herminia Diana Rosas, who can be reached at 617-726-5532 M-F 9-5.
If you have questions about the scheduling of appointments or study visits, call Ms. Sanja Nedic,
M.A. at 207-408-1763 M-F 9-5.
If you want to speak with someone not directly involved in this research study, please contact
the Partners Human Research Committee office. You can call them at 617-424-4100.
You can talk to them about:
Your rights as a research subject
Your concerns about the research
A complaint about the research
Also, if you feel pressured to take part in this research study, or to continue with it, they want to
know and can help.
If I take part in this research study, how will you protect my privacy?
During this research, identifiable information about your health will be collected. In the rest of
this section, we reIer to this inIormation simply as 'health inIormation. In general, under
federal law, health information is private. However, there are exceptions to this rule, and you
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 9 of 11
Subject Identification
should know who may be able to see, use, and share your health information for research and
why they may need to do so.
In this study, we may collect health information about you from:
Past, present, and future medical records
Research procedures, including research office visits, tests, interviews, and
questionnaires
Who may see, use, and share your identifiable health information and why they may
need to do so:
Partners research staff involved in this study
The sponsor(s) of this study, and the people or groups it hires to help perform this
research
Other researchers and medical centers that are part of this study and their ethics boards
A group that oversees the data (study information) and safety of this research
Non-research staff within Partners who need this information to do their jobs (such as for
treatment, payment (billing), or health care operations)
The Partners ethics board that oversees the research and the Partners research quality
improvement programs.
People from organizations that provide independent accreditation and oversight of
hospitals and research
People or groups that we hire to do work for us, such as data storage companies, insurers,
and lawyers
Federal and state agencies (such as the Food and Drug Administration, the Department of
Health and Human Services, the National Institutes of Health, and other US or foreign
government bodies that oversee or review research)
Public health and safety authorities (for example, if we learn information that could mean
harm to you or others, we may need to report this, as required by law)
Other:
Some people or groups who get your health information might not have to follow the same
privacy rules that we follow. We share your health information only when we must, and we ask
anyone who receives it from us to protect your privacy. However, once your information is
shared outside Partners, we cannot promise that it will remain private.
Because research is an ongoing process, we cannot give you an exact date when we will either
destroy or stop using or sharing your health information.
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 10 of 11
Subject Identification
The results of this research study may be published in a medical book or journal, or used to teach
others. However, your name or other identifying information wi ll not be used for these purposes
without your specific permission.
Your Privacy Rights
You have the right not to sign this form that allows us to use and share your health information
for research; however, iI you don`t sign it, you can`t take part in this research study.
You have the right to withdraw your permission for us to use or share your health information
for this research study. If you want to withdraw your permission, you must notify the person in
charge of this research study in writing. Once permission is withdrawn, you cannot continue to
take part in the study.
If you withdraw your permission, we will not be able to take back information that has already
been used or shared with others.
You have the right to see and get a copy of your health information that is used or shared for
treatment or for payment. To ask for this information, please contact the person in charge of this
research study. You may only get such information after the research is finished.
I nformed Consent and Authorization
Statement of Study Doctor or Person Obtaining Consent
I have explained the research to the study subject.
I have answered all questions about this research study to the best of my ability.
Study Doctor or Person Obtaining Consent Date/Time
Statement of Person Giving Informed Consent and Authori zation
I have read this consent form.
This research study has been explained to me, including risks and possible benefits (if
any), other possible treatments or procedures, and other important things about the study.
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
Partners HealthCare System
Research Consent For m
General Template
Version Date: February 2010
Page 11 of 11
Subject Identification
I have had the opportunity to ask questions.
I understand the information given to me.
Signature of Subj ect:
I give my consent to take part in this research study and agree to allow my health information to
be used and shared as described above.
Subject Date/Time
Consent Form Version:
Consent Form Title: MEG Consent am2 clear
IRB Protocol No: 2013P000356 Sponsor Protocol No: N/A
Consent Form Valid Date: 6/3/2013 IRB Amendment No: AME2 Sponsor Amendment No: N/A
IRB Expiration Date: 5/14/2014 IRB Amendment Approval Date: 6/3/2013
You might also like
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- 1 A New Science of Heat: Machines Fitted To Develop That PowerDocument9 pages1 A New Science of Heat: Machines Fitted To Develop That Powerkafka73No ratings yet
- Indoor Soccer Captains MeetingDocument14 pagesIndoor Soccer Captains Meetingkafka73No ratings yet
- Ari Jan 16 CVDocument73 pagesAri Jan 16 CVkafka73No ratings yet
- Moon Group ProjectDocument14 pagesMoon Group Projectkafka73No ratings yet
- Research PaperDocument9 pagesResearch Paperkafka73No ratings yet
- MRI Training Manual PDFDocument35 pagesMRI Training Manual PDFkafka73No ratings yet
- Kuhn (1) Scribd PDFDocument2 pagesKuhn (1) Scribd PDFkafka73No ratings yet
- (Paul Magriel) BackgammonDocument208 pages(Paul Magriel) Backgammonkafka73100% (1)
- Revolutions - Is Rather Expansive, and Thus What Follows Is Only A Brief Summarization ofDocument2 pagesRevolutions - Is Rather Expansive, and Thus What Follows Is Only A Brief Summarization ofkafka73No ratings yet
- Towards A Facsio-Sympotomatic System of MyopathyDocument1 pageTowards A Facsio-Sympotomatic System of Myopathykafka73No ratings yet
- Chess Visualization Course 1, General Tactics - Anderson, I - 2007-2011Document328 pagesChess Visualization Course 1, General Tactics - Anderson, I - 2007-2011mbti2607100% (26)
- VerticalmasteryDocument86 pagesVerticalmasteryvlada#750% (2)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Site Selection AssignmentsDocument3 pagesSite Selection AssignmentsDeepan55No ratings yet
- GABRIEL Characterizing - and - Detecting - Mismatch - in - Machine-Learning-Enabled - SystemsDocument8 pagesGABRIEL Characterizing - and - Detecting - Mismatch - in - Machine-Learning-Enabled - SystemsMarcus ViniciusNo ratings yet
- Understanding Nursing Research Building An Evidence-Based Practice 6th Edition by Susan K. Grove-Jennifer R. Gray - Nancy Burns - Test BankDocument56 pagesUnderstanding Nursing Research Building An Evidence-Based Practice 6th Edition by Susan K. Grove-Jennifer R. Gray - Nancy Burns - Test BankalishcathrinNo ratings yet
- CHRISTIANS, C. G. (2005) Ethics and Politics in Qualitative Research PDFDocument26 pagesCHRISTIANS, C. G. (2005) Ethics and Politics in Qualitative Research PDFMichele RamosNo ratings yet
- Chapter 3 Ethical Issues in Conducting Research PDFDocument26 pagesChapter 3 Ethical Issues in Conducting Research PDFAhmed Ben BellaNo ratings yet
- Title: Firstname Lastname, Firstname Lastname and Firstname LastnameDocument5 pagesTitle: Firstname Lastname, Firstname Lastname and Firstname LastnameazizahNo ratings yet
- Elgibility Criteria 7th Ed HospitalDocument2 pagesElgibility Criteria 7th Ed HospitalEng. Edelson MartinsNo ratings yet
- Informed Consent For Clinical TrialDocument16 pagesInformed Consent For Clinical TrialsidraNo ratings yet
- Research Design and Methods A Process Approach 10Th Edition Bordens Solutions Manual Full Chapter PDFDocument31 pagesResearch Design and Methods A Process Approach 10Th Edition Bordens Solutions Manual Full Chapter PDFvijayanmarylin360100% (8)
- CITIop 3Document5 pagesCITIop 3miriam tidalNo ratings yet
- RRR Call For Proposals EnglishDocument14 pagesRRR Call For Proposals EnglishIsrael Celi ToledoNo ratings yet
- Quality Assurance Journal - 2001 - Bohaychuk - Standard Operating Procedures For Clinical Research Personnel Part 9Document20 pagesQuality Assurance Journal - 2001 - Bohaychuk - Standard Operating Procedures For Clinical Research Personnel Part 9deepa_ragu5695No ratings yet
- Anda - of AndaDocument46 pagesAnda - of AndaTarikNo ratings yet
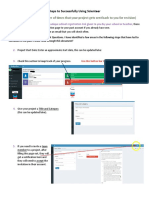
- Steps To Successfully Using ScienteerDocument6 pagesSteps To Successfully Using Scienteerapi-343437101No ratings yet
- DIV - memo.NO.350S.2019 CompressedDocument52 pagesDIV - memo.NO.350S.2019 CompressedZairene VPNo ratings yet
- Regulatory: Kalam OF WestDocument47 pagesRegulatory: Kalam OF WestSusmita GhoshNo ratings yet
- A Guide To Conduct Clinical Trials in MalaysiaDocument124 pagesA Guide To Conduct Clinical Trials in MalaysiasaraNo ratings yet
- 3 History and Principles of Good Clinical Practice 1Document24 pages3 History and Principles of Good Clinical Practice 1Larissa Miguel Severa100% (2)
- 1.approval Form 1BDocument1 page1.approval Form 1BlepoyNo ratings yet
- Experimental Psychology 7th Edition Myers Test BankDocument33 pagesExperimental Psychology 7th Edition Myers Test BankAmyWaltersjfbra100% (15)
- Research Protocol PDFDocument7 pagesResearch Protocol PDFthornapple25No ratings yet
- GXP and CGXP in Bio/Pharmaceutical Industry: Prof. Dr. Basavaraj K. NanjwadeDocument115 pagesGXP and CGXP in Bio/Pharmaceutical Industry: Prof. Dr. Basavaraj K. Nanjwadebabu1438No ratings yet
- Wcc-Mused Thesis Handbook PDFDocument12 pagesWcc-Mused Thesis Handbook PDFHilda HiewNo ratings yet
- The Belmont Report: History, Principles and ApplicationDocument10 pagesThe Belmont Report: History, Principles and ApplicationCarlos IzaguirreNo ratings yet
- Famotidine Use and Quantitative Symptom Tracking For COVID-19 in Non-Hospitalised Patients: A Case SeriesDocument6 pagesFamotidine Use and Quantitative Symptom Tracking For COVID-19 in Non-Hospitalised Patients: A Case SeriesAbdullahi Yusuf MohamedNo ratings yet
- UntitledDocument14 pagesUntitledKolawole Kolawole YUSUFNo ratings yet
- 2 Ethical Aspects of Nursing ResearchDocument35 pages2 Ethical Aspects of Nursing ResearchGrape JuiceNo ratings yet
- Ama StsDocument26 pagesAma StsJannielLintagToledo50% (2)
- A Guide To Writing A Senior Thesis in LinguisticsDocument50 pagesA Guide To Writing A Senior Thesis in LinguisticsShaza DiboNo ratings yet
- Pedagogical Ethics in Educational ResearchDocument3 pagesPedagogical Ethics in Educational ResearchWilliam MolnarNo ratings yet