Professional Documents
Culture Documents
Design of Packed Bed Absorber Column
Uploaded by
Virendra BhagatOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
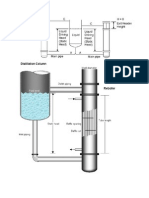
Design of Packed Bed Absorber Column
Uploaded by
Virendra BhagatCopyright:
Available Formats
Packed_Bed_absorber
PED- III
DESIGN OF PACKED BED ABSORBERSTEP 1: CALCULATE Yb, Yt , Xt
Yb = kmol of GAS ABSORBED / kmol of inerts Yt = kmol of GAS ABSORBED / kmol of inerts
(at bottom) (at top)
STEP 2: CALCULATION OF FLOW RATES:
From the V-L-E graph drawn, calculate (Lm/Gm) min (Lm/Gm)actual = (1.1 to 1.5 times) (Lm/Gm)min From (Lm/Gm)actual, calculate (Lm)actual {(Gm)actual is given in question or can be calculated from the question} Let us denote (Lm)actual = Lm and (Gm)actual = Gm Inert Mass Balance: Gm(Yb Yt) = Lm(Xb Xt) (from this eqn. calculate Xb)
Now, calculate avg. mol. Wt (at top and bottom of gas and liquid) Calculate the following values of molar flow rates (kmol/time):Gm, b = Gm(1+ Yb) Lm, b = Lm(1+ Xb) Gm, t= Gm(1+ Yt) Lm, b = Lm(1+ Xt)
From above values calculate Gb, Gt , Lb , Lt by multiplying respective avg. mol. Wts. Calculate densities, if not given in question.
STEP 3: CALCULATION OF TOWER HEIGHT:
Calculate {L/G x (g/L)1/2} for top and bottom and select the greater among the two. Now, from fig 5.33(below) we get the value of
Where, G =Superficial mass flow rate of the gas kg/s.m2 L and g=liquid and gas density, kg/m3 = liquid viscosity, cP
Packed_Bed_absorber
PED- III
The value of Fp {packing factor} is take given with packing substance details or see table. So, we substitute all the other values and obtain the value of Gas superficial velocity, Gf Gas flow rate of bottom is fixed so that the cross section can be calculated. For this we have to operate below the flooding limit, thus G we choose should be 60-85% of Gf . Then, operating velocity G = 0.80Gf C.S. area of absorber, Ac = Gb or Gt /G From here calculate the diameter of tower Dc. CHECK Dc/dp > 10 ; for satisfactory design (dp given with question)
(metre)
{depends on step-3}
STEP 4: DEGREE OF WETTING:
We have to calculate the degree of wetting rate: Lw ={L/ (Ac L a)} m3/m-s [L Lb or Lt ; depends on step-3]
(This value must be > 0.85 ft3/ft-hr)
Packed_Bed_absorber
PED- III
STEP 5: PRESSURE DROP CALCULATION: P = C2 x [10]C3Utl x g x (Utg)2
Where, P = mm H2O/m packing g = gas density ,kg/m3 C2 and C3 = constants given in question. Utg and Utl are superficial velocities of gas and liquid respectively Utg = {G / g AC} Utl = {G / l AC}
STEP 6: TOWER HEIGHT Calculations: Z = HOG x NOG
Where, Z, height of tower. HOG: height of overall gas transfer unit. NOG: number of overall gas transfer unit. HOG = HG + (mGm/Lm) HL Where, m = slope of linear equilibrium relationship.
Cornells Method:
The generalized equation of Cornell et al, for gas phase transfer units, HG
HG = (0.029 D1.11 Z0.33 Scg0.5) / (L f1 f2 f3)0.5
Dg , diffusion coefficient for mixture of gases (given) Scg = g / (g Dg)
{ will be given}
f1, f2 & f3 - given in question
L = Lt / Ac We will get an expression in the form of HG = .. x z0.33 The generalized equation of Cornell et al, for liquid phase transfer units, HL
)(
Packed_Bed_absorber
PED- III
Where, C: correction factor (0.48 for CO2 question) : Correlation parameter =liquid viscosity, Pa.s =Liquid density, kg/m3 DL =liquiddiffusion coefficient, m2/s {given} Z =height of packing, m We get the expression as HL = x z0.15 Now substitute the values of HG and HL in the equation HOG = HG + (mGm/Lm) HL in which, Gm/Lm = {(Gm/Lm)t + (Gm/Lm)b } / 2
NOG Calculations: ( )
From the graph calculate Y* and fill in the following table: Y Plot a graph of 1/(Y Y*) vs Y NOG = {Area under the curve from Yt to Yb} So we have, Height of the tower, Z = HOG x NOG Y* 1/(Y-Y*)
= [HG + (mGm/Lm)HL] NOG
Packed_Bed_absorber
PED- III
You might also like
- Design of AbsorberDocument9 pagesDesign of Absorberhaseeb tahir50% (2)
- Chap3-2e SO2 Absorption ExampleDocument8 pagesChap3-2e SO2 Absorption Exampledarkelf_riderNo ratings yet
- Design of Valve TrayDocument4 pagesDesign of Valve TrayVirendra BhagatNo ratings yet
- Mechanical design of sieve plate columnDocument65 pagesMechanical design of sieve plate columnLi Ann Chung92% (12)
- Distillation Column DesignDocument29 pagesDistillation Column Designrasithapradeep100% (4)
- Design of A Continuous Distillation Column For A Multi Component MixtureDocument45 pagesDesign of A Continuous Distillation Column For A Multi Component MixtureSerkan73% (11)
- Absorber DesignDocument26 pagesAbsorber DesignNauman Arif Waince100% (4)
- Packed column design - Calculation of packing heightDocument6 pagesPacked column design - Calculation of packing heightJeremy100% (1)
- Design of An Absorption Tower For The Separation of Acrylonitrile in - IndustryDocument9 pagesDesign of An Absorption Tower For The Separation of Acrylonitrile in - IndustryLouell Nikki HipulanNo ratings yet
- Distillation Column DesignDocument120 pagesDistillation Column DesignJaspreet Singh94% (36)
- DESIGN OF SIEVE PLATE ColumnDocument5 pagesDESIGN OF SIEVE PLATE ColumnVirendra Bhagat100% (1)
- Design of Absorption ColumnDocument33 pagesDesign of Absorption ColumnAli Hassan50% (2)
- Design of AbsorberDocument18 pagesDesign of AbsorberAdeel Ahmed100% (2)
- Gas Absorption and Gas StrippingDocument14 pagesGas Absorption and Gas StrippingDozdi86% (7)
- Packed Absorber Column DesignDocument12 pagesPacked Absorber Column Designarslan Maqbool0% (1)
- PackedColumn PDFDocument189 pagesPackedColumn PDFamadan64No ratings yet
- Absorber DesignDocument2 pagesAbsorber DesignDhrumil Gandhi100% (1)
- Distillation Column DesignDocument24 pagesDistillation Column DesignBa Tawa Na100% (1)
- Design of Distillation TraysDocument25 pagesDesign of Distillation Trayssatishchemeng100% (8)
- Distillation Column DesignDocument20 pagesDistillation Column DesignSandeep Challa100% (1)
- 8 Absorber DesignDocument16 pages8 Absorber DesignilhamriswandaaNo ratings yet
- Direct Contact Gas-LiquidDocument7 pagesDirect Contact Gas-LiquidgiovanniNo ratings yet
- 2a - Packed ColumnsDocument55 pages2a - Packed ColumnsDoaa Badarneh100% (1)
- Distillation Column DesignDocument130 pagesDistillation Column DesignKhushboo Khanna100% (5)
- Lecture 4 Reflux Ratio and Column DesignDocument13 pagesLecture 4 Reflux Ratio and Column DesignMohammedTalib100% (1)
- Aspen Plus Modeling of Distillation, Absorption and Stripping ProcessesDocument44 pagesAspen Plus Modeling of Distillation, Absorption and Stripping ProcessesWahab Maqbool100% (1)
- Packed Column Design for Chlorine Gas AbsorptionDocument5 pagesPacked Column Design for Chlorine Gas AbsorptionRyan MooreNo ratings yet
- Design of Packed Absorption Tower Using One Inch Ceramic BerlDocument86 pagesDesign of Packed Absorption Tower Using One Inch Ceramic BerlObiora IfeyinwaNo ratings yet
- Fluidized Bed ReactorDocument25 pagesFluidized Bed Reactorkennethmsoriano0% (1)
- Guidelines For Ethylene Quench Tower Rev IntroDocument12 pagesGuidelines For Ethylene Quench Tower Rev IntroGuntoro AliNo ratings yet
- Design of Distillation ColumnDocument21 pagesDesign of Distillation Columnusmanafzal246100% (1)
- Distillation Column Design ReportDocument27 pagesDistillation Column Design ReportUsama Akram100% (1)
- Packed Column Design GuideDocument64 pagesPacked Column Design GuideChelsea Martinez100% (1)
- Two Marks CH 6603 Mass TransferDocument17 pagesTwo Marks CH 6603 Mass Transfersampathkumar100% (1)
- CO2 Removal Process Design ReportDocument50 pagesCO2 Removal Process Design ReportMuhammad TauseefNo ratings yet
- Bubble Cap Plate For Distillation ColumnDocument26 pagesBubble Cap Plate For Distillation Columnsanjukec100% (2)
- Packed Columns Vs Plate ColumnsDocument8 pagesPacked Columns Vs Plate ColumnsSiri Nal100% (1)
- Distillation Column ReportDocument81 pagesDistillation Column ReportJaspreet Singh67% (9)
- Design of Packed Bed Reactor Catalyst Based On Shape, SizeDocument14 pagesDesign of Packed Bed Reactor Catalyst Based On Shape, SizeJenz Lee100% (1)
- Packed Bed AbsorptionDocument4 pagesPacked Bed AbsorptionSenthilNathanNo ratings yet
- Chemical Design of Heat Exchanger TerdesakDocument22 pagesChemical Design of Heat Exchanger TerdesakNor Ain100% (4)
- Understand Heat Flux Limitations On Reboiler Design PDFDocument10 pagesUnderstand Heat Flux Limitations On Reboiler Design PDFthawdarNo ratings yet
- Process Simulation For Distillation DesignDocument34 pagesProcess Simulation For Distillation DesignLenin PrabhuNo ratings yet
- Quench Tower (Gcu-2), ZeeshanDocument18 pagesQuench Tower (Gcu-2), ZeeshanAnkit Mehrotra100% (1)
- Book Reviews and Distillation Tray FundamentalsDocument1 pageBook Reviews and Distillation Tray FundamentalsdonyaNo ratings yet
- Adsorption Chapter: Key Concepts and EquipmentDocument77 pagesAdsorption Chapter: Key Concepts and EquipmentAkvis Fauzi RamadhaniNo ratings yet
- Distillation Column DesignDocument27 pagesDistillation Column Designl7ani100% (9)
- Thermosiphon ReboilerDocument6 pagesThermosiphon Reboilerme12m11350% (2)
- L - 17 Catalyst Deactivation: Prof. K.K.Pant Department of Chemical Engineering IIT DelhiDocument23 pagesL - 17 Catalyst Deactivation: Prof. K.K.Pant Department of Chemical Engineering IIT DelhiMehul VarshneyNo ratings yet
- Multi Component DistillationDocument94 pagesMulti Component DistillationMayur J Mehta60% (5)
- Dme BDocument8 pagesDme Bdewi xiaNo ratings yet
- (E1) Chapter 2.2 DP2 Equipment Specification SheetDocument2 pages(E1) Chapter 2.2 DP2 Equipment Specification SheetXin Yen Chia100% (1)
- Fixed-Bed Reactor Design and Diagnostics: Gas-Phase ReactionsFrom EverandFixed-Bed Reactor Design and Diagnostics: Gas-Phase ReactionsRating: 4 out of 5 stars4/5 (5)
- Process Engineering for a Small Planet: How to Reuse, Re-Purpose, and Retrofit Existing Process EquipmentFrom EverandProcess Engineering for a Small Planet: How to Reuse, Re-Purpose, and Retrofit Existing Process EquipmentNo ratings yet
- Fortran Programs for Chemical Process Design, Analysis, and SimulationFrom EverandFortran Programs for Chemical Process Design, Analysis, and SimulationRating: 4 out of 5 stars4/5 (1)
- Distillation Design and Control Using Aspen SimulationFrom EverandDistillation Design and Control Using Aspen SimulationRating: 5 out of 5 stars5/5 (2)
- Design of Packed Bed Absorber-: Step 1: Calculate Y, Y, XDocument5 pagesDesign of Packed Bed Absorber-: Step 1: Calculate Y, Y, XerenpaksoyNo ratings yet
- Chap3 2eDocument8 pagesChap3 2ehortalemosNo ratings yet
- Air Path Estimation On Diesel HCCI Engine: J. Chauvin, N. Petit, P. RouchonDocument8 pagesAir Path Estimation On Diesel HCCI Engine: J. Chauvin, N. Petit, P. RouchonalijannounNo ratings yet
- Organic Process TechnologyDocument8 pagesOrganic Process TechnologyVirendra BhagatNo ratings yet
- Liquid-Liquid ExtractionDocument8 pagesLiquid-Liquid ExtractionKheshav JhundooNo ratings yet
- Tripple Effect Evaporator Design CalculationDocument6 pagesTripple Effect Evaporator Design CalculationVirendra Bhagat100% (1)
- Equation of StatesDocument9 pagesEquation of StatesVirendra BhagatNo ratings yet
- Shell & Tube - Heat-Exchanger - DesignDocument6 pagesShell & Tube - Heat-Exchanger - DesignVirendra Bhagat100% (2)
- DESIGN OF SIEVE PLATE ColumnDocument5 pagesDESIGN OF SIEVE PLATE ColumnVirendra Bhagat100% (1)
- Design of Bubble Cap TrayDocument5 pagesDesign of Bubble Cap TrayVirendra BhagatNo ratings yet
- Conservation of Energy & MomentumDocument19 pagesConservation of Energy & MomentumVirendra BhagatNo ratings yet
- Oxidation of SO2 to SO3 favored by low temperature high pressureDocument2 pagesOxidation of SO2 to SO3 favored by low temperature high pressureVirendra BhagatNo ratings yet
- Chemical Engineering SylabusDocument34 pagesChemical Engineering SylabusVirendra BhagatNo ratings yet
- Li HongDocument4 pagesLi HongJiwen SalimNo ratings yet
- Basic Upper Preparation StepsDocument7 pagesBasic Upper Preparation StepsumidgrtNo ratings yet
- BBB BCP-15W Cycling ComputerDocument2 pagesBBB BCP-15W Cycling ComputerDannyNo ratings yet
- URA Skyline NewsletterDocument24 pagesURA Skyline NewsletterPercy Gian Marco Caceres VilcaNo ratings yet
- Hydrolysis of Lactose in Whey Permeate For Subsequent Fermentation To EthanolDocument13 pagesHydrolysis of Lactose in Whey Permeate For Subsequent Fermentation To EthanolkaltoumNo ratings yet
- Meter Moving CoilDocument4 pagesMeter Moving Coilabecdf100% (1)
- 51-11-15-001-Data Recording of Allowable Damage and Repairs-A320Document6 pages51-11-15-001-Data Recording of Allowable Damage and Repairs-A320FredNo ratings yet
- 2 13 Ionisation EnergiesDocument6 pages2 13 Ionisation EnergiesRobertLiu100% (2)
- Neem CofA 141019-4Document1 pageNeem CofA 141019-4Rahayuteuk Elf100% (1)
- Unit V Cellular Manufacturing and Flexible Manufacturing System (FMS)Document32 pagesUnit V Cellular Manufacturing and Flexible Manufacturing System (FMS)manuNo ratings yet
- DMAE Powder Safety Data SheetDocument3 pagesDMAE Powder Safety Data SheetAInhoaNo ratings yet
- Group 9 Caught in Between Modern and Contemporary ArtDocument12 pagesGroup 9 Caught in Between Modern and Contemporary Artlen lenNo ratings yet
- Ask Astrologer Report - Specific Answer To Major Concern of LifeDocument8 pagesAsk Astrologer Report - Specific Answer To Major Concern of LifecyberastroNo ratings yet
- Analysis of Caffeine Content in Kola Acuminate and Kola NitidaDocument50 pagesAnalysis of Caffeine Content in Kola Acuminate and Kola Nitidaebuka100% (1)
- The Global Commitment 2021 Progress ReportDocument39 pagesThe Global Commitment 2021 Progress ReportComunicarSe-ArchivoNo ratings yet
- C70Document3 pagesC70Jorge Luis Arevalo LopezNo ratings yet
- BE AMC-34-All Branches With IndexDocument223 pagesBE AMC-34-All Branches With IndexSrikanth RangdalNo ratings yet
- Jacques-Émile Blanche - Cahiers D'un Artiste 1916Document247 pagesJacques-Émile Blanche - Cahiers D'un Artiste 1916matwashereNo ratings yet
- Nephrology FormulasDocument3 pagesNephrology FormulasM Patel0% (1)
- FOOD PRESERVATION CHART FOR CANNING LOW-ACID & ACID FOODSDocument2 pagesFOOD PRESERVATION CHART FOR CANNING LOW-ACID & ACID FOODSjhNo ratings yet
- Ashrae Handbook 2000 CHDocument32 pagesAshrae Handbook 2000 CHKazuto NakazatoNo ratings yet
- Compro Saj 2023 - 22052023Document58 pagesCompro Saj 2023 - 22052023Ahmad FauziNo ratings yet
- 63-2003 Local Water District Franchise and Income TaxDocument2 pages63-2003 Local Water District Franchise and Income Taxapi-247793055100% (1)
- Safety Data Sheet: SECTION 1: Identification of The Substance/mixture and of The Company/ UndertakingDocument10 pagesSafety Data Sheet: SECTION 1: Identification of The Substance/mixture and of The Company/ UndertakingBradyNo ratings yet
- WDM Pon:: Systems and TechnologiesDocument27 pagesWDM Pon:: Systems and Technologiesducnm1977No ratings yet
- Kohler RRT ManualDocument40 pagesKohler RRT Manualjmh488100% (1)
- 2019-Ag-8750 Poultry Farm Visit ReportDocument7 pages2019-Ag-8750 Poultry Farm Visit ReportYOUSAF0% (1)
- Electroconvulsive Therapy: Presented By: Shweta Surwase F.Y. M.Sc. (Mental Health Nursing) MIMH, PuneDocument48 pagesElectroconvulsive Therapy: Presented By: Shweta Surwase F.Y. M.Sc. (Mental Health Nursing) MIMH, PuneShweta KateNo ratings yet
- Book 1Document330 pagesBook 1ismed123No ratings yet
- Urban green-blue grids: Hammarby SjöstadDocument5 pagesUrban green-blue grids: Hammarby SjöstadAlejandra SusaNo ratings yet